 |
 |
| Korean J Ophthalmol > Volume 27(6); 2013 > Article |
Abstract
Purpose
To evaluate choroidal thickness in diabetes patients using spectral-domain optical coherence tomography.
Methods
We examined 203 eyes of 203 diabetic participants and 48 eyes of 48 healthy controls. The choroidal thickness at the foveal lesion was measured by enhanced-depth imaging optical coherence tomography. The participants were grouped according to diabetic retinopathy grade: no diabetic change, mild-to-moderate or severe non-proliferative, or proliferative diabetic retinopathy. The study parameters included history, age, axial length, intraocular pressure, central retinal thickness, fasting glucose, and blood pressure.
Results
The subfoveal choroidal thickness was thinner in eyes with non-proliferative or proliferative diabetic retinopathy than in normal eyes (p < 0.01). However, there was no difference between eyes with non-proliferative and proliferative diabetic retinopathy or between eyes with no diabetic change and the controls. Eyes exhibiting macular edema showed no significant difference in choroidal thickness compared with eyes having normal macular contours.
The principal markers of diabetic eye disease are the breakdown of the blood-retinal barrier, retinal vasculature integrity, and hemodynamic abnormalities [1]. Clinical and experimental findings suggest that, along with retinal change, choroidal vasculopathy might also play a role in the pathogenesis of diabetic retinopathy. We further propose that it might be related to the severity of diabetic retinopathy. Various choroidal abnormalities including obstruction of the choriocapillaris, vascular degeneration, choroidal aneurysms, and choroidal neovascularization have been reported in previous studies on diabetic eyes [2-4].
Choroidal angiopathy, by contrast, has received relatively scant research attention. Until recently, the choroid could be evaluated only by indocyanine green angiography, laser Doppler f lowmetry, or ultrasound [5]. These techniques, though useful for determining vessel abnormalities or changes in the choroidal blood flow, provide no anatomical information on the retinal pigmentary epithelium or the choroidal layers. Spectral-domain optical coherence tomography (OCT) now allows for high-quality higher-resolution cross-sectional macula imaging. Additionally, enhanced-depth imaging (EDI) software enables highly reliable and reproducible measurement of choroidal thickness.
The present study, proceeding on the premise that a better clinical understanding of choroidal damage might enable a more accurate assessment of diabetic eye diseases, measured the choroidal thickness in patients with various stages of diabetic retinopathy and compared the results with those of age-matched healthy individuals.
We evaluated a series of diabetic patients at the diabetes clinic of a local hospital from October 2011 through June 2012. Healthy controls were selected from the health examination center of the same hospital. The study protocol was approved by the institutional review board of Hallym Medical Center; informed consent was obtained from all of the study participants.
Among the criteria for inclusion in this study, all of the participants were required to undergo an ophthalmologic examination, which entailed measurement of a fundus photograph, intraocular pressure, and axial length using optical biometry (Lenstar; Haag Steit AG, Koeniz, Switzerland), as well as a spectral-domain OCT examination (Spectralis; Heidelberg Engineering, Heidelberg, Germany). In order to avoid data duplication owing to symmetricity, only one eye of each participant was included in the study.
The exclusion criteria were the presence of refractive errors of more than ±4.0 diopters, cataract or media opacity rendering obtainment of reliable choroidal images impossible, a history of intraocular surgery or ophthalmic intervention, the presence of retinal pathology such as glaucoma or age-related macular degeneration, uncontrolled hypertension (>160/95 mmHg), or other systemic complications.
Retinal status was documented by means of a post-pupil-dilation fundus photograph of 45° field centered on the macula (KOWA VX-10; Kowa Optimed, Torrance, CA, USA). Blood pressure in the sitting position, fasting blood glucose, and hemoglobin A1c also were measured during the same visit, and information on diabetic duration was recorded. Retinopathy was classified according to the Early Treatment Diabetic Retinopathy Study criteria [6]. Macular edema was considered to be present if diagnosed based on the presence of macular thickening or edema, as assessed by OCT imaging. Age-matched healthy control individuals were included if their fundus photos showed no systemic disease or ophthalmic pathology.
To improve visualization of the innermost hyperreflective line of the sclera beneath the vessel layers of the choroid, we performed EDI OCT according to the previously reported method [7]. The choroid was scanned close enough to the eye to obtain an inverted image. This was then averaged over 100 scans using the automatic averaging and eye-tracking features, after which the horizontal section going directly through the center of the fovea was selected. The resultant images were viewed and measured with Heidelberg Eye Explorer software (Heidelberg Engineering). The choroid was measured from the outer portion of the hyperreflective line running from the retinal pigment epithelium to the inner surface of the sclera. The subfoveal choroidal thickness, which was perpendicular to retinal pigment epithelium layer, was measured by one of the authors (SHH) blinded to the individuals' information. Representative scans measuring the choroidal thickness were shown in Fig. 1.
Statistical analyses were performed using a commercially available statistical software package (SPSS ver. 11.5; SPSS Inc., Chicago, IL, USA). Comparative analyses of three or more groups were carried out using a one-way analysis of variance (ANOVA) followed by a post-test comparison using the Scheffe multiple test. Two groups were compared using one- and two-tailed t-test with Bonferroni's correction. The correlation analyses were evaluated on the basis of the Pearson correlation coefficient. A multiple regression analysis using the enter method was employed to investigate the influence of variable factors on choroidal thickness. A p-value less than 0.05 was considered statistically significant.
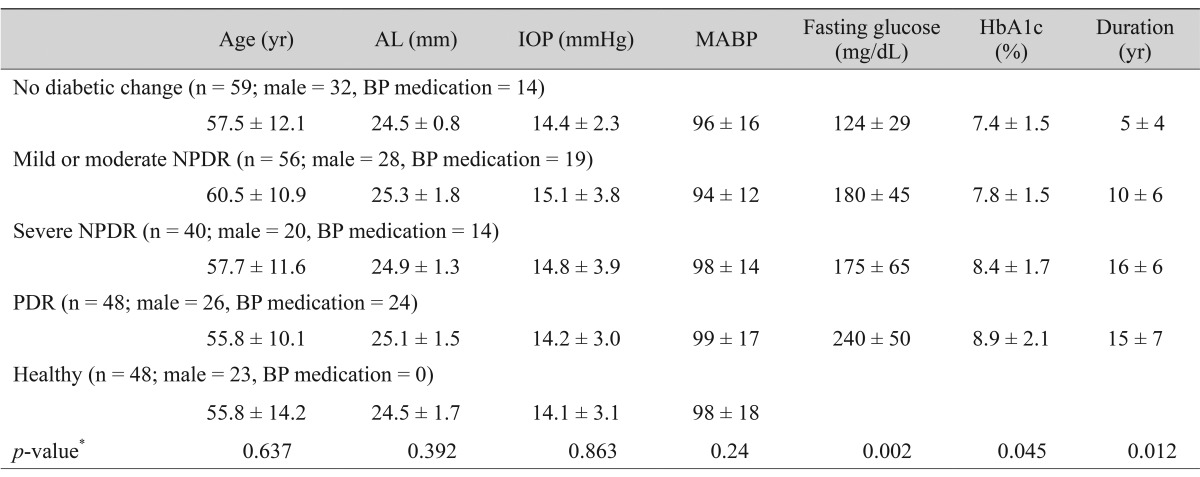
A total of 203 eyes of 203 participants (106 males and 97 females; mean age, 58.0 ± 12.0 years; range, 32 to 78 years) were enrolled for this study. The study population included 59 eyes with no diabetic change, 56 eyes with mild-to-moderate non-proliferative diabetic retinopathy (NPDR), 40 eyes with severe NPDR, and 48 eyes with proliferative diabetic retinopathy (PDR). Forty-eight eyes of 48 individuals served as controls. The study population data are listed in Table 1.
There were no differences in age, gender, axial length, intraocular pressure, or mean blood pressure among the groups (p > 0.05). The participants with diabetes, severe NPDR, and PDR showed a significantly higher fasting glucose, higher hemoglobin A1c, and longer diabetic duration (p < 0.05).
The subfoveal choroidal thickness, which was thickest among the controls, was significantly different among the groups (p < 0.001). According to a post-test analysis, there was no significant difference between the no-diabetic-change group and the controls (p = 0.846); however, a significant decrease in choroidal thickness was observed in the mild-to-moderate NPDR, severe NPDR, and PDR groups (p = 0.005, p < 0.001, p < 0.001, respectively). There were no significant differences among the mild-to-moderate NPDR, severe NPDR, and PDR groups (p > 0.05). The choroidal thickness measurement data are provided in Table 2.
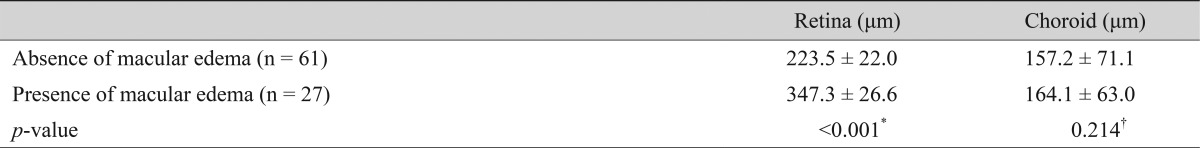
The retinal foveal thickness was significantly increased only in the severe NPDR and PDR groups compared with the controls (p < 0.001). In the no-diabetic-change and mild-to-moderate NPDR groups, there were no cases showing the presence of macular edema on OCT images. Seven (17.5%) of 40 eyes in the severe NPDR group and 20 (41.6%) of 48 eyes in the PDR group presented macular edema. Twenty-seven eyes with macular edema showed significantly decreased choroidal thickness compared with the controls (p < 0.001). However, comparisons within the severe NPDR and PDR populations revealed no significant differences in choroidal thickness between eyes with macular edema and those without (p = 0.214). The choroidal and retinal thickness measurements, as correlated with the presence of macular edema, are listed in Table 3.
The relationship between the choroidal and foveal thicknesses was also examined. In the diabetic group (R = 0.120, p = 0.127) and among eyes exhibiting macular edema (R = 0.052, p = 0.264), there were no such correlations evident.
We next analyzed choroidal thickness as related to multiple parameters including age, gender, axial length, the presence of diabetic change, fasting glucose, HbA1c, mean blood pressure, diabetic duration, and intraocular pressure. According to a multiple regression analysis, age, axial length, and grade of diabetic retinopathy were significant factors associated with choroidal thickness (R = -0.320, p = 0.01; R = -0.295, p = 0.01; R = -0.28, p = 0.031; respectively). The remaining factors showed no correlation (p > 0.05).
The possible role of choroidal vessels in the pathophysiology of diabetic retinopathy has been investigated in previous studies. It was found that similar metabolic changes affect the retinal and choroidal vascular beds, and that similar growth factors are produced and released in the diabetic choroid and retina [8-11]. It was suggested that the choroidal vascular system is affected by diabetes and might in fact be involved in the pathogenesis of diabetic retinopathy. Recently, the emergence of spectral-domain OCT has allowed for the assessment of the choroidal cross-sectional structure and its thickness [12,13]. A comprehensive understanding of choroidal change is critically important to the evaluation of diabetic eye diseases; unfortunately, information on choroidal anatomical changes in diabetic patients remains incomplete.
In the present study, individuals manifesting various stages of diabetic retinopathy were compared with healthy controls for the respective subfoveal choroidal thickness findings on OCT. The results revealed a significant decrease in choroidal thickness for the diabetic retinopathy cases. Indeed, despite the finding by McCourt et al. [14] that choroidal thinning in diabetic retinopathy was not significant after adjusting for age, the present results showed diabetic retinopathy to be significantly associated with choroidal thickness in addition to age and axial length. This is consistent with the data of previous investigations reporting choroidal thinning in diabetic eyes [5]. It has been theorized that decreased choroidal thickness might be related to retinal tissue hypoxia, as the choroid is the major source of nutrition for the retinal pigmentary epithelium and outer retinal layers [15]. Furthermore, choroidal thinning could explain the increased susceptibility to retinal hypoxia and ischemia in diabetics [16]. Finally, it was reported that choroidal blood flow in diabetes patients can be decreased with laser Doppler flowmetry and indocyanine green angiography [4,17-20]. In any case, the extent of the correlation between choroidal thickness and choroidal circulation in diabetes patients will have to await further study using new technology, such as Doppler OCT.
There have been some small pilot studies on choroidal thickness in diabetes patients. Regatieri et al. [5] reported central choroidal thinning in NPDR and treated-PDR groups as compared with healthy controls. Esmaeelpour et al. [21] also reported the same result in a no-diabetic-change group and a NPDR group and proposed that the thickness decreases in the subfoveal and inferior regions of the choroid may be related to early, preclinical changes in diabetes. Owing to the small scale and limited populations of these investigations, neither evaluated choroidal thickness changes according to all of the diabetic retinopathy grades. In the present study, there was no difference in choroidal thickness between eyes with no diabetic change and the controls, which suggests that choroidal thinning and retinal change occur at about the same time. A study of patients with no diabetic change observed decreased choroidal blood flow as the result of diabetic retinopathy [18]. We posit, therefore, that earlier choroidal circulation alteration could induce choroidal thinning at a later stage. Variability of susceptibility to choroidal circulation, in fact, might explain the above-noted different result of Esmaeelpour et al. The main differences between their and our study populations were the methods used to measure the choroid and our population's older age and longer diabetic duration (about ten years). Esmaeelpour et al. [21] used a choroidal thickness map instead of relying on single-point measurements by three dimensional 1,060-nm OCT. In addition to the method difference, we feel that age could have influenced the inconsistency observed between the two studies, particularly as older age is a demonstrated important factor related to choroidal atrophy.
Interestingly, we found that both the NPDR and PDR groups presented similar decreases of choroidal thickness compared with the controls. The progression of retinopathy from mild to proliferative change did not induce additional choroidal thinning. We believe that the functional integrity of the choroid in diabetic retinopathy is maintained after initial damage. Further, the primary surplus of choroidal vessels and blood flow might, in part, explain the relative resistance to influences of diabetic retinopathy, independent of proliferative change. Indeed, eyes exhibiting macular edema did not show more pronounced choroidal thinning than eyes without macular edema, and there was no correlation between retinal thickness and choroidal thickness at the foveal lesion. Regatieri et al. [5] reported the same findings. Thus, the development or severity of diabetic macular edema seems not to be associated with additional choroidal thinning via OCT analysis.
Systemic as well as ocular factors might also affect choroidal thickness. In an evaluation of variable factors such as blood pressure, glucose control status using fasting glucose and HbA1c, and diabetic duration, we found no effects on choroidal thickness. However, it is possible that choroidal thickness change is caused by other factors having a role in the pathogenesis of diabetic retinopathy, such as smoking or hyperlipidemia [22-24].
A strength of this study is the similarity of the groups with respect to age and axial length, which are known to affect the choroid [13,25]. The present study is also distinguished from previous reports by its larger study population. Furthermore, to minimize confounding factors, we included only naive eyes of various diabetic retinopathy grades.
There are also some limitations to the present methodology. First, the approximately 50 participants per diabetic group is a relatively small number. Second, because we measured the subfoveal choroidal thickness using the planimetric scale software on the display, the results might contain slight errors, though this is the best clinical method currently available with the current OCT equipment. Third, we did not evaluate locational changes of choroidal thickness. Lastly, we did not consider the diurnal change of choroidal thickness. Unlike retinal thickness, it has been reported that the choroid shows a diurnal variation of about 30 µm [26,27]. Additional clinical studies on larger populations are needed for a more detailed evaluation of subfoveal choroidal thickness changes.
In conclusion, the choroid thickness of subfoveal regions was significantly decreased in diabetic retinopathy patients compared with controls. The proliferative change or presence of macular edema did not result in additional choroidal thinning. Further investigation will be required before the precise role of choroidal thickness changes in the development of diabetic retinopathy can be determined.
REFERENCES
1. Cunha-Vaz J, Faria de Abreu JR, Campos AJ. Early breakdown of the blood-retinal barrier in diabetes. Br J Ophthalmol 1975;59:649-656.



2. Hidayat AA, Fine BS. Diabetic choroidopathy: light and electron microscopic observations of seven cases. Ophthalmology 1985;92:512-522.


3. Weinberger D, Kramer M, Priel E, et al. Indocyanine green angiographic findings in nonproliferative diabetic retinopathy. Am J Ophthalmol 1998;126:238-247.


4. Shiragami C, Shiraga F, Matsuo T, et al. Risk factors for diabetic choroidopathy in patients with diabetic retinopathy. Graefes Arch Clin Exp Ophthalmol 2002;240:436-442.


5. Regatieri CV, Branchini L, Fujimoto JG, Duker JS. Choroidal imaging using spectral-domain optical coherence tomography. Retina 2012;32:865-876.



6. Early Treatment Diabetic Retinopathy Study Research Group. Grading diabetic retinopathy from stereoscopic color fundus photographs: an extension of the modified Airlie House classification. ETDRS report number 10. Ophthalmology 1991;98(5 Suppl):786-806.


7. Spaide RF, Koizumi H, Pozzoni MC. Enhanced depth imaging spectral-domain optical coherence tomography. Am J Ophthalmol 2008;146:496-500.


8. Tilton RG, LaRose LS, Kilo C, Williamson JR. Absence of degenerative changes in retinal and uveal capillary pericytes in diabetic rats. Invest Ophthalmol Vis Sci 1986;27:716-721.

9. Kobayashi S, Fukuta M, Kontani H, et al. A quantitative assay for angiogenesis of cultured choroidal tissues in streptozotocin-diabetic Wistar and spontaneously diabetic GK rats. Jpn J Pharmacol 1998;78:471-478.


10. MacGregor LC, Rosecan LR, Laties AM, Matschinsky FM. Altered retinal metabolism in diabetes. I. Microanalysis of lipid, glucose, sorbitol, and myo-inositol in the choroid and in the individual layers of the rabbit retina. J Biol Chem 1986;261:4046-4051.


11. Lutty GA, McLeod DS, Merges C, et al. Localization of vascular endothelial growth factor in human retina and choroid. Arch Ophthalmol 1996;114:971-977.


12. Margolis R, Spaide RF. A pilot study of enhanced depth imaging optical coherence tomography of the choroid in normal eyes. Am J Ophthalmol 2009;147:811-815.


13. Ikuno Y, Kawaguchi K, Nouchi T, Yasuno Y. Choroidal thickness in healthy Japanese subjects. Invest Ophthalmol Vis Sci 2010;51:2173-2176.


14. McCourt EA, Cadena BC, Barnett CJ, et al. Measurement of subfoveal choroidal thickness using spectral domain optical coherence tomography. Ophthalmic Surg Lasers Imaging 2010;41(Suppl):S28-S33.


15. Linsenmeier RA, Padnick-Silver L. Metabolic dependence of photoreceptors on the choroid in the normal and detached retina. Invest Ophthalmol Vis Sci 2000;41:3117-3123.

16. Bearse MA Jr, Han Y, Schneck ME, Adams AJ. Retinal function in normal and diabetic eyes mapped with the slow flash multifocal electroretinogram. Invest Ophthalmol Vis Sci 2004;45:296-304.


17. Geyer O, Neudorfer M, Snir T, et al. Pulsatile ocular blood f low in diabetic retinopathy. Acta Ophthalmol Scand 1999;77:522-525.



18. Langham ME, Grebe R, Hopkins S, et al. Choroidal blood flow in diabetic retinopathy. Exp Eye Res 1991;52:167-173.


19. Nagaoka T, Kitaya N, Sugawara R, et al. Alteration of choroidal circulation in the foveal region in patients with type 2 diabetes. Br J Ophthalmol 2004;88:1060-1063.



20. Schocket LS, Brucker AJ, Niknam RM, et al. Foveolar choroidal hemodynamics in proliferative diabetic retinopathy. Int Ophthalmol 2004;25:89-94.


21. Esmaeelpour M, Povazay B, Hermann B, et al. Mapping choroidal and retinal thickness variation in type 2 diabetes using three-dimensional 1060-nm optical coherence tomography. Invest Ophthalmol Vis Sci 2011;52:5311-5316.


22. UK Prospective Diabetes Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. BMJ 1998;317:703-713.



23. Yang L, Gong H, Wang Y, et al. Nicotine alters morphology and function of retinal pigment epithelial cells in mice. Toxicol Pathol 2010;38:560-567.


24. Li XQ, Larsen M, Munch IC. Subfoveal choroidal thickness in relation to sex and axial length in 93 Danish university students. Invest Ophthalmol Vis Sci 2011;52:8438-8441.


25. Fujiwara T, Imamura Y, Margolis R, et al. Enhanced depth imaging optical coherence tomography of the choroid in highly myopic eyes. Am J Ophthalmol 2009;148:445-450.


Fig. 1
Optical coherence tomography images using enhanced-depth imaging. The choroidal thickness (white arrow) was defined as the vertical distance from the hyperreflective line of the Bruch's membrane to the outermost hyperreflective line, as measured in micrometers. (A) Representative scan of a normal individual. (B) Representative scan of an individual with severe non-proliferative diabetic retinopathy; note that the choroid is thinner than in the normal individual. (C) Representative scan of an individual with proliferative diabetic retinopathy (PDR). The image shows normal macular contours. (D) Representative scan of an individual with PDR and macular edema.

- TOOLS
-
METRICS

- Related articles
-
Assessment of Scleral Contour Changes in High Myopia by Optical Coherence Tomography2022 April;36(2)




 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



