Systemic Factors Associated with Central Serous Chorioretinopathy in Koreans
Article information
Abstract
Purpose
To investigate systemic factors associated with central serous chorioretinopathy (CSC).
Methods
We retrospectively reviewed the medical records of 113 Korean patients who were diagnosed with CSC and who underwent history taking with a specialized questionnaire for CSC. They were matched for age and gender at a ratio of 1 : 3 to 339 normal controls. Normal controls were consecutively selected from a database at the Health Promotion Center. General characteristics and medical histories were compared between the two groups. The statistical analyses used included independent t-test, Mann-Whitney test, Fisher's exact test, and multivariate logistic regression analysis.
Results
There were 90 men and 23 women in the CSC group, and the male-female ratio for both groups was 3.9 : 1. The mean age of the patients was 45.6 years. In multivariate analysis, hypertension (odds ratio [OR], 2.327; 95% confidence interval [CI], 1.349-4.013), use of medicinal plants (OR, 2.198; 95% CI, 1.193-4.049), sleep disturbances (OR, 1.732; 95% CI, 1.096-2.739), and snoring (OR, 1.727; 95% CI, 1.058-2.820) were strongly associated with CSC.
Conclusions
Hypertension, sleep disturbance, snoring, and medicinal plant use were identified as factors associated with CSC. Expanded history taking, including systemic factors and culture-specific behavior related to stress or fatigue such as use of medicinal plants, will be helpful in identifying Korean patients at an increased risk for CSC.
Central serous chorioretinopathy (CSC) is characterized by idiopathic neurosensory retinal detachment or retinal pigment epithelial (RPE) detachment at the posterior pole [1]. It was originally thought to be a disorder of the RPE; however, it is now widely accepted that the disease originates from choroidal hyperperfusion [2-6]. Recent advances in optical coherence tomography (OCT) allow for measurements of the choroid and have revealed a thicker than normal choroid in patients with CSC. Increased hydrostatic pressure in the thickened choroid seems to lead to pigment epithelial and retinal detachments [7].
The pathophysiology of choroidal thickening in CSC remains unknown. Choroidal vessels have been suggested to be under the control of the autonomic nervous system; therefore, systemic factors may influence choroidal hyperperfusion. Investigation regarding systemic findings in patients with CSC could help us to better understand the pathophysiology of CSC. Several systemic parameters such as male gender, psychological stress, and corticosteroid medication use have previously been suggested to be correlated with CSC [1,8-12]. Recently, the use of psychopharmacologic medications, which could possibly be used as a proxy for psychological stress, was suggested as a possible risk factor of CSC [13]. However, this has not been extensively investigated in Asians [14].
We retrospectively evaluated 113 patients with idiopathic CSC collected from the database of the Health Promotion Center and compared them with a normal control group matched 1 : 3 for age and gender.
Materials and Methods
Approval for this study was obtained from the institutional review board. All research and data collection adhered to the tenets of the Helsinki agreement. Retrospective reviews were performed on all patients who were diagnosed with idiopathic CSC at our institute between April 2009 and December 2009. We included patients who underwent fluorescein angiography and history taking with a specialized CSC questionnaire which was adopted from the questionnaires used in the Health Promotion Centers at our institution for history taking regarding systemic risk factors. The questionnaire included questions about height, weight, history of systemic and ophthalmic diseases, current medication including medicinal plants, snoring, and sleep disturbances. Snoring was documented when subjects reported snoring more than three to four times per week or when the subject's snoring was reported to bother other people. Regarding sleep disturbances, subjects were questioned whether they awoke feeling tired or if they experienced fatigue during the day.
CSC was defined as localized neurosensory retinal detachment or RPE detachment associated with a focal leak or fluorescein leakage at the level of the RPE on fluorescein angiography. OCT had been performed to confirm the presence of active disease. Chronic CSC was defined as symptoms persisting longer than six months, accompanied by a sensory retinal detachment or RPE detachment associated with diffuse fluorescein leakage. Indocyanine green angiography was performed in eyes with chronic CSC. Atypical CSC was defined as multiple serous detachments of the RPE and bullous sensory retinal detachment, dependent neurosensory detachment, epithelial tracts, diffuse RPE decompensation, subretinal deposits of fibrin and lipids, or CSC associated with secondary choroidal neovascularization (CNV). We excluded patients with evidence of associated uveitis, polypoidal choroidovasculopathy, or CNV that could have been a cause of exudation. We also excluded CSC due to steroid use.
To determine systemic factors associated with CSC, the patients were matched for age and gender at a ratio of 1 : 3 to a normal control group who had been examined in the Health Promotion Center at our institution. The normal control group was consecutively selected by reviewing charts and fundus photographs from the database of the Health Promotion Center from April 2009. We excluded controls with a history of ophthalmic diseases such as glaucoma, uveitis, or retinal disorders. We also excluded controls with abnormal findings on fundus photography.
Statistical analysis was performed using independent t-test, Mann-Whitney test, Fisher's exact test, and multivariate logistic regression analysis in SPSS ver. 12.0 (SPSS Inc., Chicago, IL, USA). Results were considered statistically significant if the p-value was less than 0.05.
Results
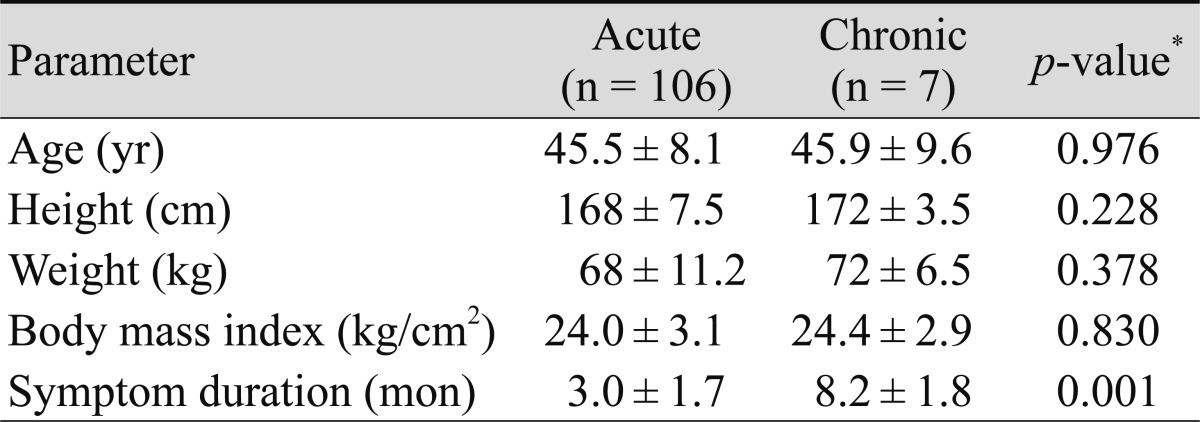
One hundred thirteen patients with CSC and 339 controls were recruited for this study. The mean age of the cases was 45.6 years, and the mean follow-up period was 7.6 months. The male-female ratio was 3.9 : 1. There were no statistically significant differences in height, weight, or body mass index (BMI) between the CSC group and the control group (Table 1). Of the 113 cases, seven had chronic CSC, and 22 had atypical CSC. There were no statistically significant differences in age, height, weight, or BMI between the subgroups of CSC patients (Tables 2 and 3).
Hypertension was more frequent in patients with CSC (25.7%) than it was in the control group (13.3%, p = 0.003, Fisher's exact test) (Table 4). Medicinal plant use occurred more frequently in patients with CSC (18.6%) than it did in the control group (13.3%, p = 0.031). Frequently used medicinal plants included unknown mixed herbal medicine, steamed red ginseng, acanthopanax, and licorice root. Some subjects reported using an herbal soup mixed with deer antlers or Sang Hwang mushrooms (Phellinus ssp). Snoring (34.5%) and sleep disturbances (44.2%) were also reported more frequently in patients with CSC compared to the controls (20.6%, p = 0.004; 28.9%, p = 0.004).
Using a multivariate binary logistic regression analysis, hypertension (odds ratio [OR], 2.327; 95% confidence interval [CI], 1.349-4.013; p = 0.002), medicinal plant use (OR, 2.198; 95% CI, 1.193-4.049; p = 0.012), sleep disturbances (OR, 1.732; 95% CI, 1.096-2.739; p = 0.019), and snoring (OR, 1.727; 95% CI, 1.058-2.820; p = 0.029) were strongly associated with CSC.
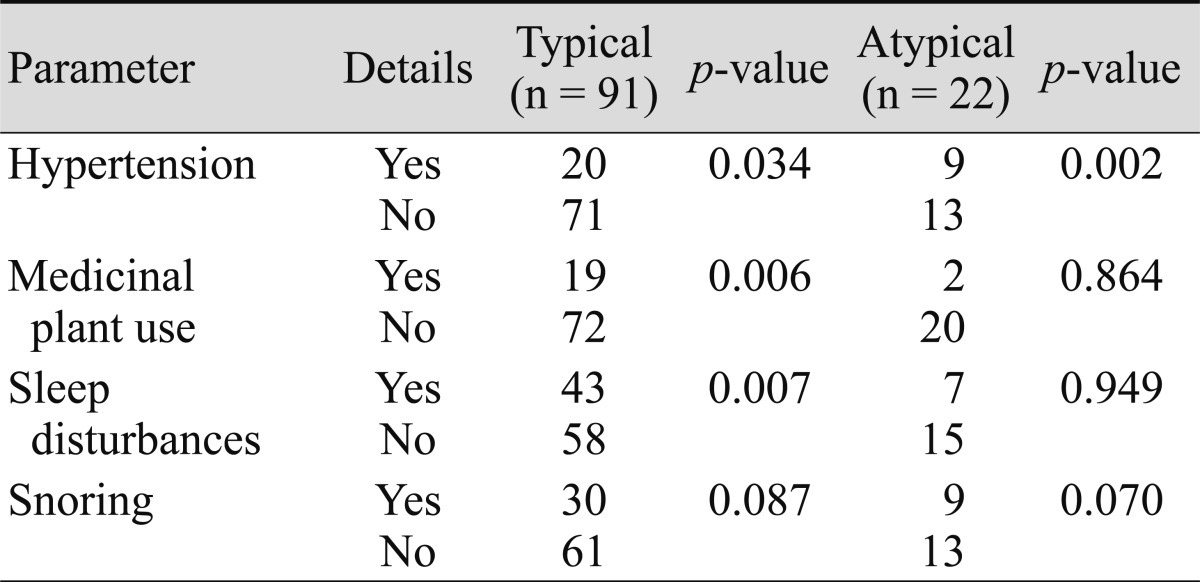
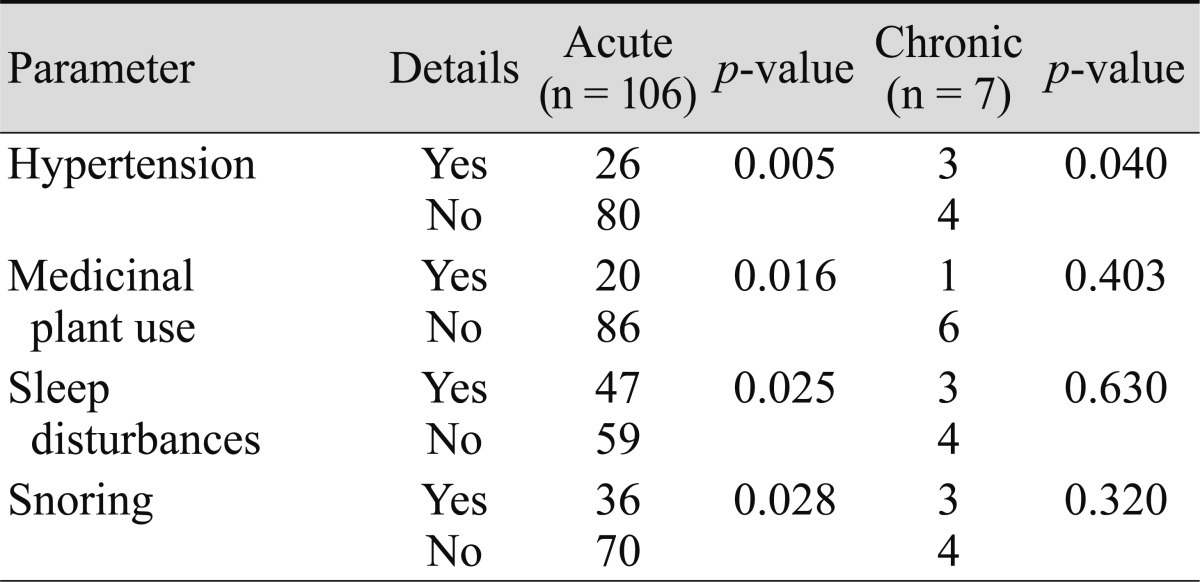
Using a multivariate logistic regression analysis of subgroups from the CSC group, the development of acute CSC was associated with hypertension, medicinal plant consumption, sleep disturbances, and snoring; typical CSC was associated with hypertension, medicinal plant consumption, and sleep disturbances. However, chronic CSC and atypical CSC were only associated with hypertension (Tables 5 and 6).
Multinomial logistic regression analysis of acute and chronic central serous chorioretinopathy subgroups (n = 113)
Discussion
In this study, the mean age of patients with CSC and the gender ratio were similar to those of previous Western studies [15]. Previous studies have reported that hypertension is a systemic risk factor for CSC; our results corroborate this finding [13,16]. Additionally, other possible systemic factors were found to be associated with CSC in this study.
A relationship between sleep apnea and CSC has been suggested in previous Western studies, although these studies were limited by either a small number of cases or a non-comparative design [17,18]. Our study had a larger number of cases and a control group; we also found that sleep disturbances and severe snoring appear to be correlated with CSC. The altered catecholamine levels and sympathetic activity that often accompany sleep disturbances may influence the onset of CSC [19,20]. Alternatively, stress, which has been acknowledged as a risk factor for CSC, might also predispose one to sleep disturbances [9].
Medicinal plants have been used for primary healthcare needs in some Asian and African countries [21-23]. Traditional medicinal plants, also known as herbal medicines, refer to the medicinal products of plant roots, leaves, and seeds that can be used to promote general health and to treat disease. Many Asian cultures have used medicinal plants not for specific illnesses, but rather for the promotion of general health [24-26]. In the present study, consumption of medicinal plants was significantly more frequent in patients with CSC compared to the control group. Some medicinal plants could produce ocular side effects [27,28]. However, the results of the current study did not directly indicate that the medicinal plant itself influences the development of CSC. Many Koreans use these traditional medicinal plants to treat fatigue [29,30]. When considering that medicinal plants are frequently sought by Korean patients because of fatigue, the seeking behavior itself may serve as a proxy for the presence of fatigue. A positive correlation between fatigue and CSC was reported in previous studies [31-38]. It was also shown in a previous Western study that patients with CSC were more likely to use pharmacotherapeutic drugs than were control subjects [13]. Tittl et al. [13] suggested that the more frequent use of psychopharmacologic medications represented the increased psychological stress in CSC patients. However, the use of psychopharmacologic medications was not different between the CSC patients and controls in our study with Koreans. Such a discrepancy may be attributed to the different control groups between the two studies. However, it could also be explained by the different cultural backgrounds of the patient groups. Koreans prefer medicinal plants to psychopharmacologic medications for relief of stress or fatigue, while Western people tend not to [27].
CSC is a self-limiting disease. However, some patients may develop a chronic form of the disease and experience severe vision loss. There are severe forms of CSC which have a poor prognosis if they are not treated. These include multiple serous detachments of the RPE and bullous sensory retinal detachment, dependent neurosensory detachment, epithelial tracts, diffuse RPE decompensation, subretinal deposits of fibrin and lipids, and CSC associated with secondary CNV [39]. The development of acute CSC in our cases was strongly associated with hypertension, medicinal plant consumption, sleep disturbances, and snoring; typical CSC was associated with hypertension, medicinal plant consumption, and sleep disturbances. However, chronic CSC and atypical CSC were only associated with hypertension.
The prevalence of hypertension in Koreans aged 40 to 49 years was 22.7% in 2009 [40]. In the present study, the prevalence of hypertension in the control group was 13.3%. This difference was due to the fact that the questionnaire used in this study was answered based on a subject's past experience with diagnosis of hypertension before examination in the Health Promotion Center or ophthalmic clinic. Indeed, among the 339 control subjects, 74 (21.8%) were diagnosed with hypertension after the examination. This prevalence is similar to that of Koreans aged 40 to 49 years (22.7%). Therefore, the practical prevalence of hypertension in CSC patients may be higher than that reported in the questionnaire (25.7%).
There are some limitations to this study. Medical records were retrospectively reviewed, and systemic factors were analyzed retrospectively. In particular, standard medical history charts rely on a subject's recall at a particular time. We did not quantitatively measure sleep disturbances or snoring, and we only included cases of idiopathic CSC. We did not accept questionnaires from patients with CSC associated with steroid use. This prevented an analysis regarding an association of corticosteroid use with CSC. As this study was a cross-sectional comparative study using medical history charts, the period and course of the disease process could not be evaluated.
In conclusion, hypertension, sleep disturbance, snoring, and medicinal plant use were identified as factors associated with CSC. Expanded history taking, including systemic factors and culture-specific behavior related to stress or fatigue such as use of medicinal plants, will be helpful in identifying Korean patients at an increased risk for CSC.
Notes
Presented in part at the 103rd meeting of the Korean Ophthalmological Society, April 3-4, 2010, Busan, Korea.
No potential conflict of interest relevant to this article was reported.