A Comprehensive Analysis of the Influence of Temperature and Humidity on Dry Eye Disease
Article information
Abstract
Purpose
To investigate the effects of humidity and temperature on dry eye disease (DED)
Methods
A retrospective, clinic-based study was conducted on DED patients undergoing dry eye treatment. Patients were followed up at least twice, and symptoms and signs were evaluated using the Symptoms Assessment Questionnaire in Dry Eye (SANDE) score, tear secretion, tear film breakup time (TBUT), ocular staining score, and tear osmolarity. Mean humidity and temperature values for 1 week before ocular examinations were used as the environmental exposure level. The relationship between humidity and temperature, with DED clinical parameters was analyzed in single- and multi-environmental factor models.
Results
The study included 33 patients with a mean age of 53.9 ± 12.2 years. The low humidity group showed significantly higher SANDE scores (p = 0.023) and tear osmolarity (p = 0.008), and the low temperature group had higher SANDE scores (p = 0.004), ocular staining scores (p = 0.036), and tear osmolarity (p < 0.001). In the linear mixed model, single factor analysis showed that an increase in humidity resulted in decreased SANDE scores (p = 0.043), and an increase in temperature led to a decrease in SANDE score (p = 0.007), ocular staining score (p = 0.007), and tear osmolarity (p = 0.012). In the multifactor analysis, changes in humidity had no significant effect on dry eye parameters, but an increase in temperature was significantly correlated with decreased SANDE score (p = 0.026), ocular staining score (p = 0.024), and tear osmolarity (p = 0.002).
Conclusions
Lower temperature led to aggravated symptoms and signs of DED and the effect of temperature on DED was more pronounced than humidity. Tear osmolarity was the most sensitive clinical parameter to be affected by climate factors in DED patients.
Dry eye disease (DED) is a common ocular surface disorder estimated to affect around 8.0% to 30.3% of the Korean population [1,2]. It is a multifactorial disease characterized by an unstable tear film, in which tear hyperosmolarity, ocular surface inflammation, and neurosensory abnormalities play etiological roles [3]. Subsequent symptoms of fluctuating visual acuity and ocular discomfort affect patients’ quality of life and pose a major public health issue.
Common risk factors of DED include older age, female sex, Asian race, and meibomian gland dysfunction [3]. Other than personal risk factors, environmental factors such as humidity and temperature are also known to be associated with dry eye prevalence, symptoms, and signs [4,5]. Specifically, low humidity has repeatedly been shown to be associated with higher dry eye prevalence [6–9]. In controlled experimental settings, lower relative humidity was reported to result in faster tear evaporation, shorter tear film breakup time (TBUT), and more ocular surface staining [10–13]. Moreover, the usage of moisture goggles was reported to ameliorate dry eye symptoms and reduce tear evaporation rate [14]. However, in real-world conditions and outside of laboratory settings, the effect humidity has on individual dry eye parameters has not yet been extensively studied. In addition, the role of temperature, one of the most important environmental factors, in dry eye is inconclusive. Some researchers suggested that higher ambient temperature was associated with more dry eye diagnosis [6,8,9], while others reported lower temperature correlated with better [15] or worse dry eye symptoms [16]. In experimental settings higher ambient temperature led to increased tear evaporation [17], but its effect on TBUT was conflicting [17,18]. Room temperature is known to be positively associated with ocular surface temperature [19] and ocular surface temperature is reported to be lower in evaporative dry eye [20]; however, the causal relationship is unclear, and the effect of environmental outside temperature on dry eye is yet to be investigated.
Domestic data regarding the influence of temperature and humidity on DED is limited. Often, clinicians tend to underestimate the impact of environmental factors, as DED treatment primarily focuses on routine artificial tears and other therapeutics. Highlighting the significance of the environment’s effects on the ocular surface, the recent TFOS Lifestyle Report [5] underscores the importance of considering these factors with patient treatment. To address this gap and gain a deeper understanding, we conducted a follow-up study on the same patients to assess how changes in humidity and temperature affect DED. This investigation involved analyzing climate data from the Korea Meteorological Administration along with clinical data obtained from a university hospital, to study the effects of real-world temperature and humidity on DED clinical parameters. Furthermore, we sought to identify which specific clinical parameters are more sensitive to these fluctuations.
Materials and Methods
Ethics statement
This retrospective study adhered to the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board of Gachon University Gil Medical Center (No. GBIRB2023-173). The requirement for informed consent was waived due to the retrospective nature of the study.
Study design and setting
The study included dry eye patients who visited the clinic from February 2019 to August 2021. To avoid confounding due to geographical factors, only patients living in Incheon Metropolitan City, Korea, who did not move during the study period were included in the study. Dry eye diagnosis was based on the guidelines of the Korean Corneal Diseases Study Group [21]. Dry eye was diagnosed when the patient had dry eye symptoms and showed at least one objective dry eye sign. Dry eye symptoms included ocular symptoms such as dryness, discomfort, foreign body sensation, or pain, and visual symptoms such as blurring or vision fluctuation. Objective signs included TBUT ≤10 seconds or positive ocular surface staining of Oxford grade ≥1. All patients used the 0.05% cyclosporine twice daily and artificial tears were used as needed. Patients with contact lens wear, usage of glaucoma eyedrops, or those with other ocular surface diseases or systemic rheumatic diseases that could affect the ocular surface were excluded from the study.
Clinical assessment of dry eye parameters
All patients underwent clinical examination by an experienced cornea specialist (DHK) to assess dry eye symptoms and signs at least twice during follow-up. Dry eye symptoms were assessed using the Symptom Assessment Questionnaire in Dry Eye (SANDE) score [22]. Tear secretion was evaluated using the Schirmer test without anesthesia, and TBUT was measured as the time between the last eye blink and the time of tear breakup upon fluorescein instillation. Ocular staining score (OSS) was recorded using the Oxford grading scale [23], and tear osmolarity was measured with TearLab (TearLab Corp) according to the manufacturer’s manual.
Data collection of environmental factors
Humidity and temperature data of Incheon Metropolitan City were collected from the Ministry of Environment of Korea on dates from February 2019 to August 2021. Data were acquired from the monitoring station closest to the subjects’ addresses. The mean values of temperature and relative humidity for 1 week before the ocular examinations were used as the environmental exposure level because environmental factors during the week preceding the examination had the most significant impact on dry eye signs and symptoms in a previous study [24].
Statistical analysis
For each patient, the dry eye parameters at the lowest humidity value were assigned to the low humidity group, while those at the highest humidity value were assigned for the high humidity group. The same logic was followed for the low and high temperature groups. To evaluate the differences in dry eye parameters between the low and high humidity or temperature groups, a paired t-test was conducted. Additionally, the effects of temperature or humidity changes on dry eye parameters were examined using a linear mixed model. This analysis was performed for both single- and multi-environmental factor models. In the single factor model, the effects of temperature and humidity were evaluated independently, while the multifactor model adjusted the temperature and humidity simultaneously to account for their potential interaction. Differences between sex were additionally analyzed. All data are presented as mean ± standard deviation, and all statistical analyses were performed using IBM SPSS ver. 20.0 (IBM Corp). A p-value of less than 0.05 was considered statistically significant.
Results
Data from 33 patients were collected in the study. The mean age was 53.9 ± 12.2 years, and 24 patients (72.7%) were female. The mean follow-up period was 15.0 ± 3.2 months and the number of follow-ups was 2.2 ± 0.4. Mean SANDE score was 65.8 ± 27.1, mean TBUT was 4.0 ± 1.4 seconds, mean OSS was 1.0 ± 0.7, mean tear secretion was 10.5 ± 7.0 mm, and mean tear osmolarity was 303.9 ± 20.6 mOsm/L. Mean relative humidity was 67.9% ± 11.0%, and mean temperature was 14.7 ± 9.8 °C. Details of baseline demographics are presented in Table 1. Fig. 1 illustrates the relationship between relative humidity and temperature. There was a significant positive correlation between humidity and temperature (r = 0.528, p = 0.002).
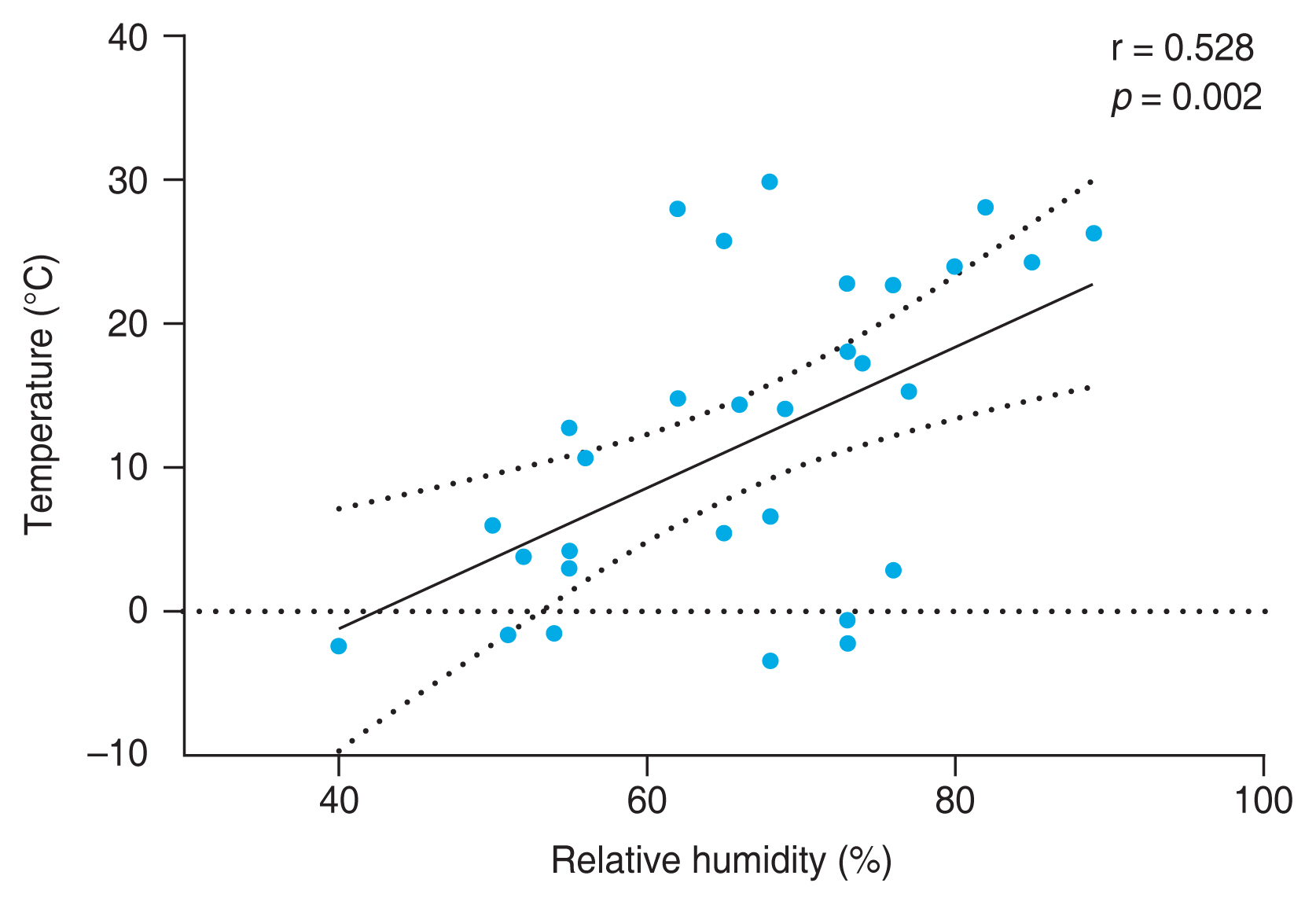
Relationship between relative humidity and temperature. There was a significant positive correlation between relative humidity and temperature (r = 0.528, p = 0.002).
Tables 2 and 3 illustrates the dry eye parameter differences between the lowest and highest humidity or the lowest and highest temperature. The mean relative humidity was 59.0% ± 7.2% in the low humidity group and 72.9% ± 8.1% in the high humidity group. SANDE score (p = 0.023) and tear osmolarity (p = 0.008) were significantly higher in the low humidity group. There were no significant differences in TBUT, OSS, and tear secretion between the low and high humidity groups. In terms of temperature, the mean temperature was 6.0 ± 7.3 °C in the low temperature group and 18.4 ± 7.3 °C in the high temperature group. SANDE score (p = 0.004), OSS (p = 0.036), and tear osmolarity (p < 0.001) were significantly higher in the low temperature group. TBUT and tear secretion were not significantly different among the two temperature groups.
In the linear mixed model using single environmental factor analysis, 1% increase in relative humidity led to a significant decrease in SANDE score (p = 0.043) and other parameters showed no significant association (Table 4). A 1 °C increase in temperature led to a significant decrease in SANDE score (p = 0.007), OSS (p = 0.007), and tear osmolarity (p = 0.012). In a multi-environmental factor analysis considering both humidity and temperature together, humidity change did not significantly affect dry eye parameters. As for temperature, 1 °C increase in temperature was associated with a significant decrease in SANDE score (p = 0.026), OSS (p = 0.024), and tear osmolarity (p = 0.002). Of the five DED parameters, tear osmolarity was the most sensitive to changes in temperature and humidity (Tables 2–4).

Effects of environmental factors on dry eye disease using single- and multi-environmental factor analyses (n = 33)
Differences between sex were also analyzed using the multi-environmental analysis model (Table 5). For both men and women, relative humidity change was not significantly associated with changes in dry eye parameters. Men exhibited a significant decrease in SANDE score (p = 0.015), ocular staining (p = 0.009), and tear osmolarity (p = 0.007) along with temperature increase, whereas women only experienced changes in SANDE score (p = 0.025) and tear osmolarity (p = 0.036) according to temperature change.
Discussion
This study showed that increase in temperature was associated with alleviation of dry eye symptoms, less ocular surface staining, and lower tear osmolarity. While lower relative humidity was associated with worse symptom scores in the single factor analysis, other objective clinical parameters were not significantly affected, and the association became insignificant when using the multi-environmental model. Moreover, symptoms score and tear osmolarity were found to be consistently affected by changes in temperature regardless of analysis model or sex, and tear osmolarity was the most sensitive clinical parameter to be affected by the climate factors.
The relative lack of association between relative humidity and dry eye parameters was unexpected, considering the generalized idea from previous research that lower humidity aggravates dry eye. In experimental settings where asymptomatic patients were exposed to low relative humidity of 5% for 2 hours, ocular surface staining significantly increased and TBUT significantly decreased compared to before the exposure [11]. When Sjögren syndrome–associated dry eye patients were exposed to this condition, tear osmolarity and surface staining significantly increased [25]. In another experiment of asymptomatic patients who were exposed to shorter 1 hour exposure to 5% relative humidity, tear evaporation increased and TBUT significantly decreased [12]. Many more experiments with similar results exposed patients to desiccating stress under controlled environmental conditions, and relative humidity of desiccating stress ranged from 5% to 25% [13,26,27]. In our study the mean relative humidity was 67.9% ± 11.0%, and the minimum value was 40%, which is often the value used as the normal control condition [10,12]. It is possible that above a certain value of relative humidity, clinical parameters might not be significantly affected, which could be the reason for lack of correlation in our results. Moreover, since patients were on dry eye treatment, they may have been less affected by changes in humidity. However, in the linear mixed model of single environmental factor analysis decreased humidity was associated with worse SANDE scores, indicating patient symptoms may be affected, unlike objective parameters. That subjective symptoms worsen with lower humidity is consistent with aforementioned previous studies [6,7].
That higher temperature is associated with better clinical parameters are in line with some previous research [16,17]. A Taiwanese study [16] reported that in low ambient temperature was associated with higher ocular surface disease index (OSDI) score, similar to our study in which SANDE scores were higher with lower temperature. In an experiment using a controlled environmental chamber in which only temperature varied [17], mean TBUT increased significantly from 7.3 seconds at 5 °C to 12.4 seconds at 25 °C, and lipid layer thickness was also significantly higher at higher temperatures. While this study did not investigate ocular surface staining and our results did not reveal a significant correlation of temperature with TBUT, it can be inferred that lower temperature has a negative effect on the tear film, which might be related to the worse staining scores with lower temperature in our results. The effects of lower ocular surface temperature due to lower environmental temperature might have affected the pathophysiology of the ocular surface. When the ocular surface and eyelid temperature is below 33 °C meibum in glands was found to be thicker, impeding its normal delivery to the ocular surface [28], and ambient temperature below 10 °C was reported to result in ocular surface temperature of less than 32 °C [17]. Therefore, it is possible that in our study lower environmental temperature might have negatively affected the tear film dynamics, leading to worse symptoms and more staining.
To date, we could not find any studies on the effects of temperature on tear osmolarity. Our results showed that tear osmolarity was not only significantly higher with lower temperature, but it is also the most sensitive marker of DED to climate change. Tear osmolarity is an established diagnostic marker of DED, and increase in tear osmolarity reflects an increase in DED severity [29]. Other clinical parameters sometimes have poor correlation with subjective symptoms, which may be due to the multiple etiologies of DED and the specificity of each clinical test to one certain etiology [30]. In previous research comparing the objective clinical parameters of dry eye, tear osmolarity was found to be a superior predictor of dry eye severity [29,31]. In one study of 130 subjects [29], tear osmolarity had stronger statistical correlation with DED severity compared to TBUT and tear clearance. In another multicenter study of 314 subjects [31], tear osmolarity had superior diagnostic performance compared to TBUT, ocular surface staining, Schirmer test, and meibomian gland grading, with the highest area under the receiver operating characteristic curve. In our study, SANDE score and tear osmolarity were found to be consistently affected by temperature change regardless of analysis model or sex, suggesting that changes in tear osmolarity may be a precursor for DED symptoms. We believe that tear osmolarity may hold promise as a potential biomarker for DED in climate change.
Interestingly, the effects of temperature on DED parameters varied between men and women. While increased temperature correlated with decreased SANDE scores, OSS, and tear osmolarity in men, for women only SANDE scores and tear osmolarity were affected. It is known that due to sex steroids and other hormones, prevalence of DED is higher in women [32]. In previous studies investigating sex differences in DED symptoms and signs, women had higher OSDI scores, and less correlation between signs and symptoms [33,34]. In one study [35] women were more likely to show corneal staining compared to men, and in other studies [33,34] women had higher tear osmolarity, lower TBUT, along with higher staining scores. However, little is known about whether there are sex differences in the effects of temperature on the ocular surface. Since women did not exhibit changes in OSS along with temperature change, there may be differences in the way the ocular surface is affected between the men and women, in a mechanism that needs further investigation.
There are several limitations to our study. First, the relatively small sample size of DED patients (n = 33) may restrict generalization of the results to a larger population. Second, the relatively small number of follow-ups limited our ability to observe long term trends longitudinally within the same patient. Future studies should include a larger cohort with longer follow-up, and an additional cross-sectional approach would also be needed to assess DED parameters uniformly under consistent temperature and humidity levels across subjects. Additionally, while this study included patients undergoing the same topical treatment of twice daily 0.05% cyclosporine eyedrops, variable patient usage of artificial tears as needed could have impacted patient symptoms and ocular surface erosion status in subjects. Moreover, subjects may be more affected by indoor temperature or humidity than outside environmental exposure depending on their occupation and time spent outdoors. While this study was meant to investigate the influence of environmental humidity and temperature, in future investigations, we will incorporate such factors and if possible, adjust for indoor parameters for more detailed analysis. Furthermore, we only focused on humidity and temperature in this report; however, there are other meteorological (wind and sun exposure) and pollution-related environmental factors that could also play significant roles in DED, and taking these factors into account in future investigations would offer a more comprehensive understanding of the multifaceted influence of the environment on DED [5,24,36–38]. While these limitations underscore the need for further research, our results provide insights into the effect real-world temperature and humidity have on individual clinical DED parameters, emphasizing the significance of considering environmental factors in patients with dry eye disease.
In conclusion, this study showed that lower temperature is associated with worse dry eye symptoms and more ocular surface staining, along with higher tear osmolarity values. Tear osmolarity may be a sensitive indicator of DED to temperature change. Further research is needed to explore the potential impact of other environmental hazards such as air pollutants (ozone, nitrogen dioxide, and particulate matter), as they may be associated with DED [7,24,36].
Acknowledgements
None.
Notes
Conflicts of Interest
None.
Funding
This work was supported by the Korea Environment Industry & Technology Institute (KEITI) through the Core Technology Development Project for Environmental Diseases Prevention and Management (No. 2022003310001), funded by the Ministry of Environment of Korea.



