Assessment of Perinatal Clinical Characteristics, Perinatal Risk Factors, and Microbial Profile in Congenital Nasolacrimal Duct Obstruction in a Tertiary Care Center: A Descriptive Study
Article information
Abstract
Purpose
To assess the perinatal risk factors, clinical characteristics, and microbial profile in congenital nasolacrimal duct obstruction (CNLDO).
Methods
A prospective observational study was conducted in pediatric patients of age <5 years. After a thorough clinical evaluation, the diagnosis of the CNLDO was confirmed by a Jones dye test or fluorescein dye disappearance test. A microbiological culture of lacrimal sac resurge was done with a sterile swab stick without touching the lid margins. Antibiotic susceptibility was then performed for the standard antibiotics.
Results
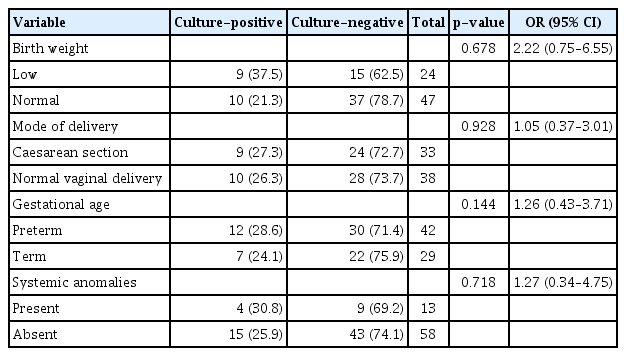
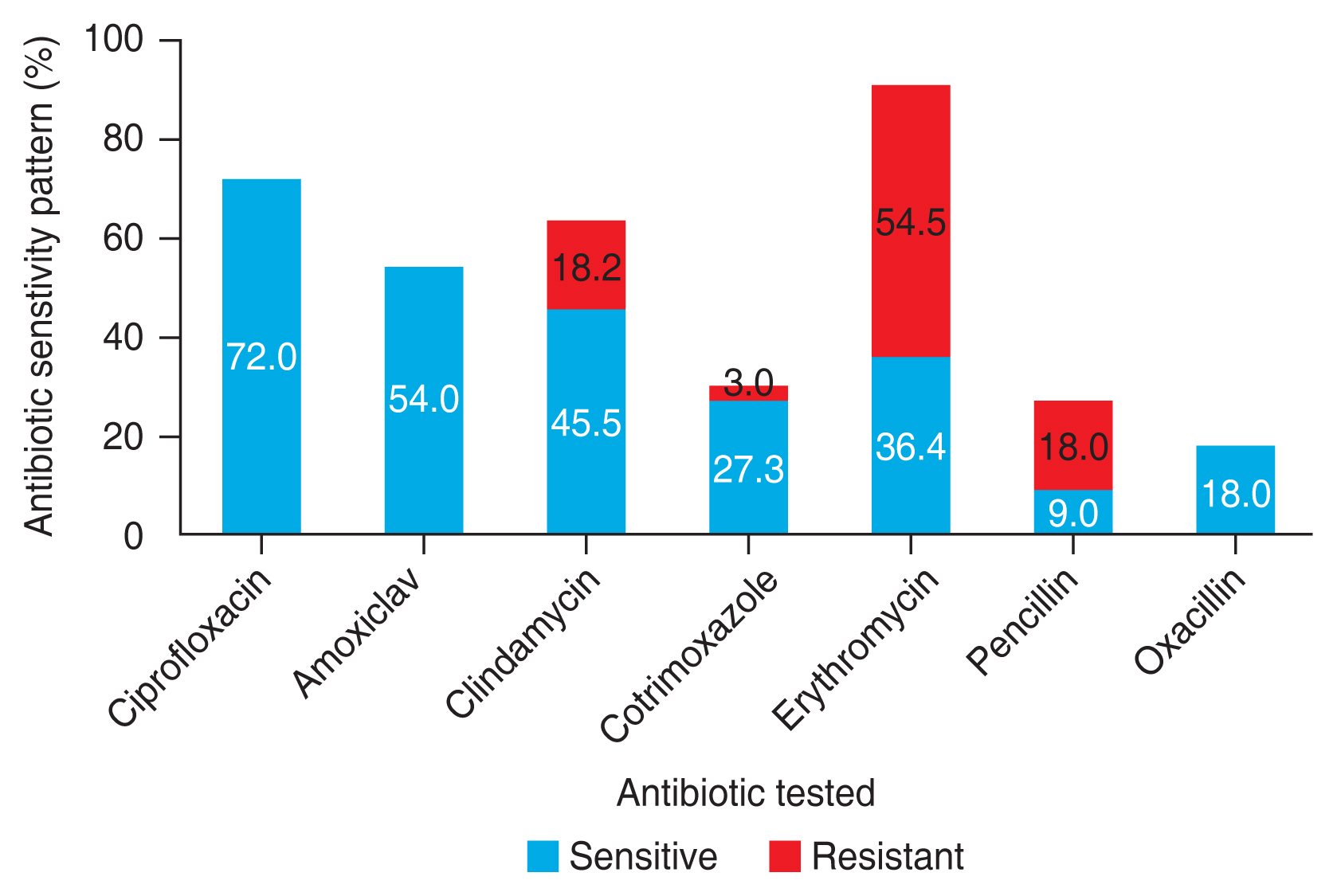
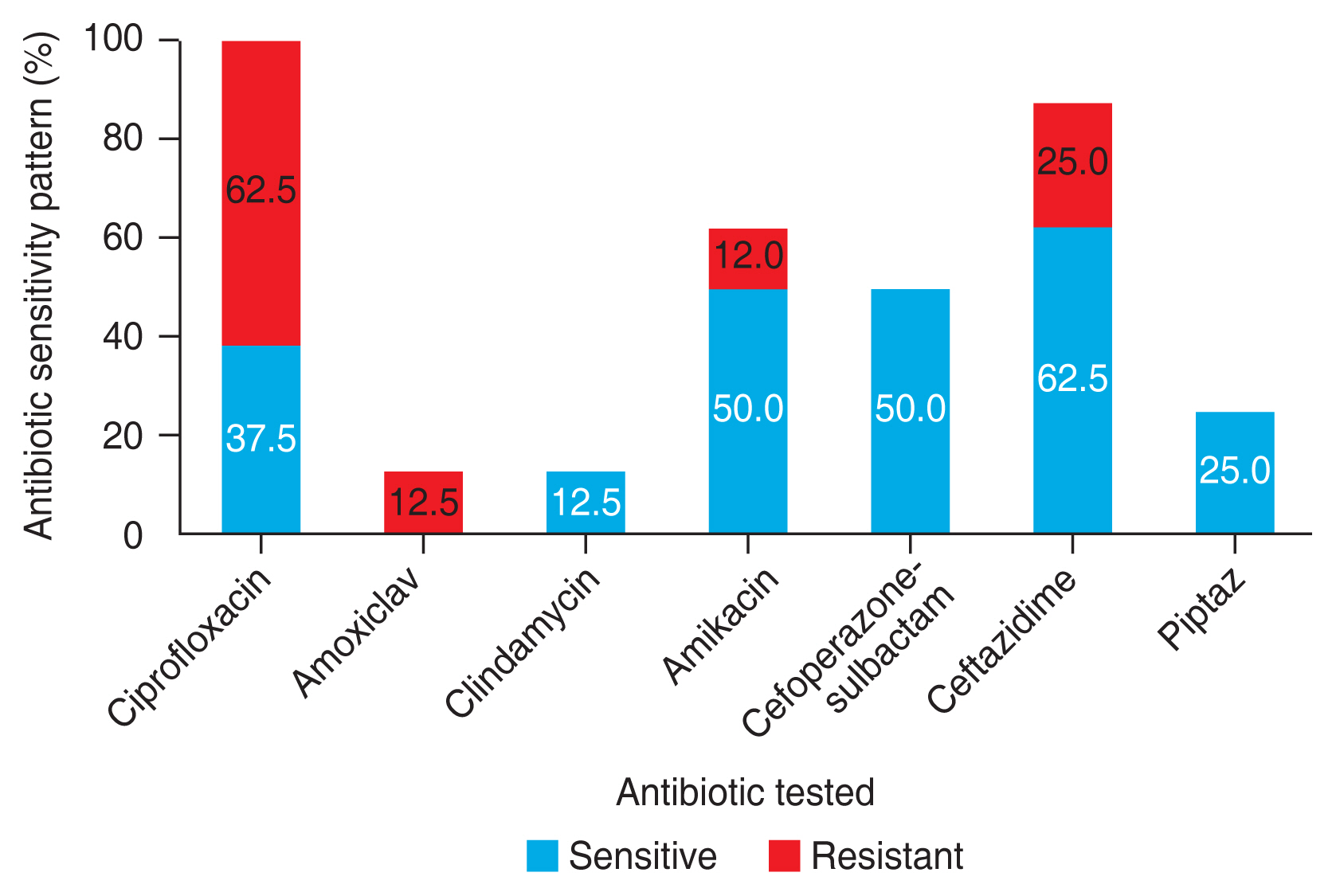
Seventy-one eyes of 52 children were included in the study. The mean age was 3.4 months. Our research found an equal number of cases born via spontaneous vaginal delivery and Caesarean section. Nine children (17.3%) had associated systemic and ocular anomalies. The number of cultures that were positive for any growth was 19 (27%). The most common isolate was Streptococcus pneumoniae which constituted eight cases (42%), followed by Pseudomonas aeruginosa (15.8%), and Escherichia coli (10.5%). Other organisms that grew were Klebsiella pneumoniae, Staphylococcus aureus, Staphylococcus epidermidis and nonfermenting gram-negative bacilli. Gram-positive organisms were susceptible to ciprofloxacin, amoxiclav, and oxacillin, and most of them were resistant to erythromycin. In comparison, gram-negative organisms showed 62.5% resistance and 37.5% susceptibility to ciprofloxacin.
Conclusions
CNLDO was more commonly unilateral, in male, preterm, and normal birth weight infants. Bilateral CNLDO was more commonly associated with coexisting ocular or systemic anomalies. Prematurity, delivery by Caesarean section, and presence of congenital anomalies were associated with a prolonged course. S. pneumoniae was the predominant isolate in our patient population.
Congenital nasolacrimal duct obstruction (CNLDO) accounts for about 6% of all newborn infants [1]. The exact etiology and predisposing factors are unknown. It occurs secondary to a delay in maturation of the lacrimal system that results in a persistent, membranous obstruction of the nasolacrimal duct. The condition is usually unilateral but may be bilateral. Infants with nasolacrimal duct obstruction typically present within the 1st month of life with epiphora, recurrent periocular crusting, or both. CNLDO most commonly is an isolated condition, but sometimes, it may be more frequent in children with Down syndrome or craniofacial anomalies [2]. The associations with refractive error and amblyopia are controversial. Obstruction of the nasolacrimal duct can lead to stagnation of fluid within the lacrimal sac area, which can get secondarily infected. Studies have shown that microbiological testing in CNLDO may have a value in predicting the obstruction [3]. Also, analyzing the lacrimal sac microorganisms and their antibiotic susceptibility in infants can prevent the complications and inappropriate use of antibiotics leading to antibiotic resistance [4].
Materials and Methods
A descriptive study was conducted in a tertiary care hospital between October 2018 and March 2021, after obtaining the approval from the Institutional Ethics Committee of Jawaharlal Institute of Postgraduate Medical Education and Research (No. JIP/IEC/2018/440). Written informed consents for publication of the research details and clinical images were obtained from the patients’ legal guardians. Pediatric patients of age <5 years, presenting with clinical features suggestive of CNLDO (such as epiphora, discharge with matting of lashes, swelling, and redness noticed since or shortly after birth) were included. Patients with acquired CNLDO due to trauma or nasal surgery, prior antibiotic treatment in the last 7 days, excessive tear production from a non-CNLDO induced etiology, such as allergic conjunctivitis and congenital glaucoma, with coexisting ocular conditions like conjunctivitis, blepharitis, uveitis, were excluded. Demographic data such as age, sex, and laterality were noted. Thorough birth history of antenatal, perinatal risk factors such as mode of delivery, birth weight, prematurity, exposure to teratogenic drugs or radiation, and infections in the mother was obtained from medical records available at the time of presentation or elicited via detailed history taking. All patients underwent thorough clinical assessment starting with the general examination to look for facial malformations and syndromic features. The ocular examination included identification and ruling out other local causes of tearing such as foreign body, conjunctivitis, buphthalmos, or blepharitis. The diagnosis of the CNLDO was confirmed by a fluorescein dye disappearance test or by a Jones dye test. A microbiological culture of lacrimal sac resurge was done. The samples were obtained by applying gentle pressure over the lacrimal sac region and collected with a sterile swab stick without touching the lid margins. Following this, refraction with 1% cyclopentolate eye drops and a dilated fundus examination was performed. The samples were incubated for 2 days, and bacterial isolates were identified. Antibiotic susceptibility was then performed for the standard antibiotics using the disc diffusion method, the minimum inhibitory concentration method. Data was entered using excel and analyzed using IBM SPSS ver. 21.0 (IBM Corp., Armonk, NY, USA). Continuous variables like age and duration of symptoms were summarized as mean with standard deviation or median with a range based on the distribution of data. Categorical variables like sex, symptoms, duration of symptoms, gestational age, birth weight, mode of delivery, family history, and exposure to antenatal drugs and/or infections were summarized as frequency and percentages. Outcome variables like culture positivity, most common organism isolated, and antibiotic susceptibility were expressed as frequency and percentages. The association of the duration of symptoms and culture positivity rate with other categorical variables was carried out by using chi-square test or Fisher exact test. The statistical analysis was performed at a 5% level of significance, and a p-value of less than 0.05 was considered significant.
Results
Clinical characteristics
A total of 71 eyes of 52 children were included in the study. Most of the patients (n = 44, 84.6%), were less than 1 year of age at presentation and the mean age at diagnosis was 5.6 ± 6.6 months. Male patients constituted 33 cases (63.5%) and female patients constituted 19 cases (36.5%). Among them, 33 cases (63.5%) were unilateral and 19 cases (36.5%) were bilateral. It was found that patients with systemic anomalies had a higher frequency of bilateral cases but with no statistical significance (p = 0.588). Most of the children (n = 38, 73%) had tearing of eyes associated with ocular discharge. One case had swelling of the lacrimal sac area associated with redness of the eye. (Fig. 1A–1D and Table 1).
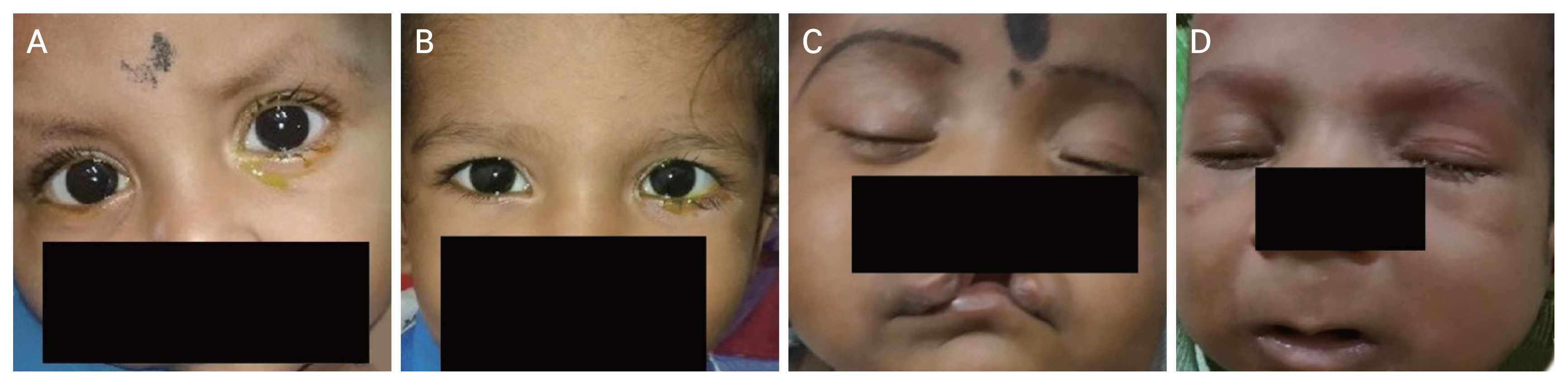
Clinical photographs showing patients of congenital nasolacrimal duct obstruction with (A, B) a positive fluorescein dye disappearance test in the left eye, (C) a cleft lip, and (D) Pierre Robin sequence. Written informed consents for publication of the clinical images were obtained from the patients’ legal guardians.
Risk factors
Our study found an equal number of cases born via spontaneous vaginal delivery and Caesarean section (CS). A history of prematurity was noted in 30 cases (57.7%). Among them, a majority were late preterm children. It was also seen that 65% of preterm children had symptoms for more than 6 months duration as opposed to 35% of the term children (p = 0.339). Sixteen cases (30%) belonged to the low birth weight category. A family history of CNLDO was present in three cases (6%). Nine cases (17.3%) had associated systemic and ocular anomalies. Our study found three cases (5.8%) that had a history of maternal infection. All three had a history of maternal genital tract infection. Regarding the associated systemic and ocular anomalies, the commonly found anomalies were Pierre Robin sequence, mucopolysaccharidosis, craniosynostosis, Down syndrome, cleft lip and palate, and telecanthus. Of these children, four had bilateral disease, and 44.4% of the patients had symptoms more than 6 months duration as opposed to 37.2% of the cases that had no associated systemic anomalies, although there was no statistical significance (Table 2). Analysis of the cycloplegic refraction showed no significant difference in the refractive error of both eyes in cases with unilateral CNLDO.
Microbiological profile
The number of cultures that were positive for any growth was 19 (26.8%). The cases that had bilateral disease grew the same organisms except for three cases where one eye showed no growth. Risk factors such as prematurity and vaginal delivery seem to have been associated with an increased percentage of culture positivity rate. However, the results are not statistically significant (p = 0.144 and p = 0.928, respectively) (Table 3). Gram-positive organisms constituted about 11 cases (58.0%), and the most common isolate was Streptococcus pneumoniae which constituted about nine cases (47.4%), followed by Staphylococcus aureus (5.3%), and Staphylococcus epidermidis (5.3%). The most common gram-negative organism identified as Pseudomonas aeruginosa, which constituted three cases (15.8%), followed by Escherichia coli (10.5%), Klebsiella pneumoniae (10.5%), and nonfermenting gram-negative (5.3%). Gram-positive organisms were fully susceptible to ciprofloxacin, amoxiclav, and oxacillin, and most of them were resistant to erythromycin. Gram-negative organisms were susceptible to cefoperazone-sulbactam and piptaz, with 62.5% resistance and 37.5% susceptibility to ciprofloxacin (Fig. 2, 3).
Discussion
CNLDO is a common ocular condition characterized by excessive tearing of eyes with or without mucoid discharge, secondary to the obstruction due to blockage in the nasolacrimal duct pathway [5]. Our study showed 33% bilateral cases, like most studies showing bilateral affection in one-third of the cases. In a retrospective study on CNLDO patients, it was found that patients with Down syndrome predominantly had bilateral disease, and the most common presentation was tearing [6]. This might be attributed to the fact that anatomical abnormalities in Down syndrome are either focal stenosis or diffuse stenosis, other than obstructions at its distal end. Our study also found a higher frequency of bilaterality in cases with coexisting systemic and ocular anomalies. The median age group of the patients was 4 months which correlates with the natural course of congenital disease. Most of the patients in our study had tearing associated with mucopurulent discharge than tearing alone, similar to the results of a study by Sathiamoorthi et al. [7], correlating with obstruction at the valve of Hasner, the most common site of obstruction, causing mucopurulent discharge than tearing alone. Our study found an equal number of cases born via normal vaginal delivery (NVD) and CS. Several studies found a significant association between CNLDO and CS, stating that the valve of Hasner opens because of increased uterine pressure during labor and by the heightened activity of collagenolytic enzymes released after rupturing of membranes [8–10]. This contrasts with a study by Valcheva et al. [9], which concluded that CS significantly reduces the risk for the development of CNLDO. However, they added that not dividing CS as primary and secondary to be a limitation to this finding. Tavakoli et al. [8], in a study comparing CS and NVD, found that cases born via CS did not show spontaneous resolution and required surgical intervention. Our study did not follow up the patients for spontaneous resolution but found an increase in the duration of symptoms in cases born via CS (>6 months in 55%) than cases born via NVD (>6 months in 47%), suggesting a prolonged course of the disease in the group born by CS. A history of prematurity was noted in 30 cases (56%). Among them, a majority were late preterm children. It was also seen that 65% of preterm children had symptoms for more than 6 months duration as opposed to 35% of the term children. Several studies have reported a higher incidence of CNLDO in premature infants. While the membrane of the lacrimal puncta gets canalized by the 7th month of gestational age, the membrane near the lower end of nasolacrimal duct has a persistent membrane most of the time, explaining why premature infants have a higher incidence of CNLDO. Al-Faky et al. [11], in their study on 200 premature and full-term infants, also found a higher percentage of CNLDO in premature children when compared to full-term infants. Sixteen cases (30%) belonged to the low birth weight category. While several studies found a significant association between low birth weight and CNLDO, most of our patients had a birth weight that was appropriate for age. However, Valcheva et al. [9] in their study found that prematurity and low birth weight decrease the risk for CNLDO but could not explain this association.
The role of genetics has not been thoroughly studied in CNLDO. It is reported to be a sporadic or multigenic mode of inheritance [12]. A study by Valcheva et al. [9] could not find a hereditary predisposition for CNLDO. Our study found an associated family history of CNLDO in three cases (6%). A study by Aldahash et al. [13] on the risk factors for developing CNLDO reported 19 patients (14.7%) who had a first-degree relative with CNLDO and concluded a significant association between CNLDO and family history (p < 0.001). They also found that found infection during pregnancy to be a statistically significant risk factor for developing CNLDO. However, age of the mother, exposure to X-rays, drug intake during the first trimester, smoking status were not found to be significantly associated with the development of CNLDO [13]. Our study found only three cases (5.8%) that had a history of maternal genital tract infection. About nine cases (17.3%) had associated systemic and ocular anomalies. The commonly found anomalies were Pierre Robin sequence, mucopolysaccharidosis, craniosynostosis, Down syndrome, cleft lip and palate, and telecanthus. While Down syndrome and ectrodactyly ectodermal dysplasia-cleft syndrome have been reported to be most associated with CNLDO [14], our study had two cases with Pierre Robin sequence and only a single case with Down syndrome. Few studies in the literature establish an association of cleft lip and congenital bony obstruction of the nasolacrimal duct where the cleft lip deformity has been found to influence the nasolacrimal duct diameter without affecting the nasolacrimal duct length [15,16]. We also found an increased duration of symptoms in the group that had associated systemic and ocular anomalies. There have been studies reporting an association of unilateral CNLDO and anisometropic amblyopia. The reason that has been postulated is that constant watering and discharge cause distortion of the perceived image, which may hinder the process of emmetropization [17]. Another proposed association between dacryostenosis and hyperopic anisometropia was attributed to anatomical orbital factors such as a smaller orbit or tight nasolacrimal passage in conjunction with a short hyperopic eye. But, with a retrospective study design, it is difficult to establish whether CNLDO was present first or risk factors for amblyopia were present first, and hence a causal relationship of these two variables cannot be established [18].
Our study found a culture positivity rate of 27% (n = 19) obtained from 71 specimens. This frequency is a little less when compared to several other studies. Al-Faky et al. [11] showed a culture positivity rate of 87.9% from 181 lacrimal duct specimens and Madan et al. [1] found a culture positivity rate of 85.2% from 121 specimens. The lower positivity rate in our study might be since most of the cases were a referral from an outside hospital with a prior history of antibiotic usage. Also, though most patients had mucopurulent discharge than isolated tearing, not all turned out to be culture-positive. The source of microbes can be the normal conjunctival flora, the upper respiratory tract, or the genital tract of the mother. Hence, the fact that not all discharges are infective must be kept in mind when a decision on whether to start antibiotics arises. Our study showed a predominant growth of the gram-positive organism, S. pneumoniae. A clinical study by Kuchar et al. [19] showed a culture positivity rate of 72.64% from 50 lacrimal sacs resurge samples. The predominant organism identified as S. pneumoniae, followed by Haemophilus influenzae, and the most drugs with high sensitivity for microbes were identified to be ofloxacin and tetracycline. Another study conducted in Wardha by Madan et al. [1] on CNLDO showed similar results with Haemophilus influenzae and S. pneumonia to be the most common microbial isolates followed by Moraxella catarrhalis similar to other studies, which also showed ciprofloxacin, chloramphenicol, bacitracin, and neomycin to be the most effective local antibiotics for these microorganisms [4,20,21]. The most common gram-negative organisms identified as P. aeruginosa and E. coli have been predominant organisms in dacryocystitis in acquired or adults than in CNLDO [21,22]. Gram-positive organisms showed high rates of sensitivity to ciprofloxacin, and a majority of them were resistant to erythromycin. This correlates with a study that mentioned a 40% resistance to erythromycin and thus ineffective in CNLDO [4]. While gram-negative organisms were moderately susceptible to ciprofloxacin.
Nayak et al. [23] in their study on 146 patients with concurrent microbial keratitis and CNLDO, found that stasis of tears in the lacrimal sac causes a change in the virulence pattern of commensal organisms making them more pathogenic leading to poor prognosis of keratitis. Identification of the organism and treatment with appropriate antibiotics can help to avoid local infectious changes that lead to intrasaccular stenosis and poor surgical outcome. Also, studies have found that certain microorganisms are associated with tighter obstructions of the nasolacrimal duct than others which might have an implication on the treatment outcome, making identification of the microbiota yet another useful tool [11].
Strengths of this study include its prospective nature, restricting to the pediatric population, and the analysis of the clinical and microbiologic features. Limitations of this study include a small sample size, affecting the level of statistical significance, the short duration of the study, the possibility of contamination of culture specimen with ocular flora, and factors influencing the outcome of study like prior antibiotic usage and recall bias.
CNLDO, though a benign and self-remitting condition, must have a uniform treatment protocol as to when to start the antibiotics as a part of conservative management. Inclusion of cases where a standard technique of collecting lacrimal sac aspirate specimens was followed, such as cases whose samples were collected in the operating room just prior to the surgical management under sterile precautions, might give an accurate picture of the actual microbial spectrum. Also, conducting further studies with a large sample size in the same locality and usage of a wide range of culture mediums and antibiotic susceptibility techniques for identifying organisms might give a better insight into the prevailing microbial pathogen. The natural course of the disease must be assessed with a regular follow-up of the cases treated conservatively rather than assessing the duration of disease to establish an accurate association with the risk factors and the severity of CNLDO. For further study, a multicentered investigation with a large sample size, the inclusion of both symptomatic and asymptomatic cases by screening and following standard specimen collecting techniques must be performed.
In conclusion, CNLDO was more common in male, preterm, and normal birth weight infants. It was commonly unilateral, and bilateral cases were associated with coexisting anomalies. Prematurity, delivery by CS, and the presence of congenital anomalies were associated with a prolonged course. S. pneumoniae was the predominant isolate in our patient population.
Acknowledgements
None.
Notes
Conflicts of Interest: None.
Funding: None.