 |
 |
| Korean J Ophthalmol > Volume 36(4); 2022 > Article |
|
Abstract
Purpose
Mucormycosis is a multisystemic, aggressive, and an opportunistic fungal infection. The most common type is rhino-orbito-cerebral mucormycosis (ROCM) accounting for almost 40% of the cases. In this study, we analyzed the etiopathology, clinical features, treatment outcome, and the factors associated with outcome in ROCM.
Methods
Case records of 52 patients of ROCM were analyzed. Clinical parameters, laboratory parameters, imaging findings, treatment regime, and treatment outcome details were retrieved from each case record. The outcome measures were evaluated as treatment success and treatment failure. Univariate and logistic regression analyses were performed to identify factors associated with treatment outcome.
Results
On univariate analysis, factors associated with poor treatment outcome were uncontrolled blood sugar, blood urea, diabetic ketoacidosis, duration of ocular symptoms, no perception of light at presentation, ptosis, conjunctival chemosis, relative afferent pupillary defect, two or more sinus involvement, bony erosion of orbital wall, intracranial extension, intraconal invasion, etc. On subsequent logistic regression analysis, factors that maintained significant association with poor treatment outcome were uncontrolled blood sugar (adjusted odds ratio [aOR], 1.17; p = 0.018), no perception of light at presentation (aOR, 10.67; p = .006), ptosis at presentation (aOR, 3.90; p = 0.03), conjunctival chemosis (aOR, 7.11; p = 0.024), relative afferent pupillary defect (aOR, 10.60, p = 0.01), central retinal artery occlusion at presentation (aOR, 3.54; p = 0.021) and two or more sinus involvement (aOR, 4.90; p = 0.009).
Mucormycosis is a multisystemic, aggressive and an opportunistic fatal fungal infection caused by the fungi Mucormycetes and class Mucorales. The common forms of mucormycosis are rhino-orbito-cerebral (ROC), pulmonary, cutaneous, gastrointestinal, disseminated, and isolated renal mucormycosis. Among these, the most common type is ROC mucormycosis (ROCM) accounting for almost 40% of the cases [1-3]. The most common route of infection is by invasion from the adjacent paranasal sinuses or direct traumatic inoculation into the orbit. After the fungal colonization in the sinonasal mucosa, it enters the orbit through the ethmoid and maxillary sinuses or via the nasolacrimal duct, and spread towards the brain occurs either through ophthalmic artery, superior orbital fissure, or via cribriform plate. The spread of the infection to the frontal lobes and to the cavernous sinus occurs via perivascular and perineural channels through the cribriform plate and the orbital apex [4,5].
ROC disease is seen almost exclusively in immunocompromised hosts. Approximately 60% to 80% of the cases occur in patients with diabetic ketoacidosis [6-9]. In this retrospective study, we analyzed the etiopathology, clinical features, and treatment outcome of patients with ROCM attending a tertiary care hospital. We also analyzed the factors affecting the final outcome with an emphasis on the ocular features that can predict the outcome.
The study was conducted after the approval from the Institutional Ethics Committee of the Jawaharlal Institute of Postgraduate Medical Education and Research, Puducherry, India (No. JIP/IEC/2020/183), and it adhered the ethical principles mentioned in the Declaration of Helsinki. Written informed consent for publication (including the images) has been obtained from the parent of the patient.
We conducted a retrospective study of the case records of ROCM patients admitted from January 2019 to April 2020. Case record details including demographic history, medical history, treatment details, laboratory parameters, and imaging findings were all retrieved for each patient. Ocular evaluation details retrieved for each patient included best-corrected visual acuity (BCVA), intraocular pressure, slit-lamp examination findings, and dilated fundus examination findings. Diagnosis of mucormycosis was established for each patient based on the clinical evaluation, imaging (contrast-enhanced computed tomography [CECT]), histopathological examination, and microbiological assessment of the samples. ROCM was categorized into three distinct stages based on the clinical features and level of disease extension. Stage I disease was defined as isolated sinonasal disease. Stage II disease had isolated rhino-orbital disease: difficulty in vision, ocular movement restriction, proptosis, external/internal ophthalmoplegia, central retinal artery occlusion (CRAO), etc. Stage III had ROCM disease, with cerebral involvement indicated by cavernous sinus thrombosis, altered sensorium, or hemiplegia [10]. Inclusion criteria were stage II and stage III disease. Isolated sinonasal disease (stage I) cases were excluded from our study as this stage had no ocular clinical features at presentation.
On clinical diagnosis of mucormycosis, treatment with systemic amphotericin B was started in all the patients. Based on the clinical staging and nature of the surgery that the patients had undergone subsequently, patients were categorized into three different treatment groups: A, B, and C. Treatment A regimen included medical treatment and nasal debridement; B regimen included medical treatment and nasal debridement with orbital exenteration; and C regimen involved only medical treatment. Patients with rhino-orbital disease (stage II) underwent either treatment A or B while patients with ROC disease (stage III) and those who were not willing for any surgery received treatment C regimen.
The outcome measures for our study were evaluated as treatment success and treatment failure. Treatment success was characterized by systemic stability, metabolic control, and disease-free condition. Treatment failure was characterized by disease progression to a severe condition, deterioration of the physical condition, or death due to the infection [10].
The data were entered and analyzed using IBM SPSS ver. 26.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were represented using mean ┬▒ standard deviation for continuous variables. Nonparametric variables were analyzed using the chi-square and Fischer exact test. Comparison of means was performed using Student t-test for parameters showing normal distribution and Mann-Whitney U-test for nonnormally distributed parameters. Normality of distribution was assessed for all continuous variables using Shapiro-Wilk test. Linear regression analysis was performed on continuous variables including blood urea, random blood sugar, creatinine, age, and duration of symptoms to assess their impact on final outcomes. Significant variables on univariate analysis were further subjected to logistic regression analysis after performing multicollinearity test. A p-value of less than 0.05 was considered statistically significant.
A total of 52 patients of mucormycosis with orbital or ocular involvement presented to Jawaharlal Institute of Postgraduate Medical Education and Research during the study period. Two patients had bilateral involvement. Thirty-six patients (69.2%) were male and 16 patients (30.7%) were female. Mean age at presentation was 51.84 ┬▒ 14.06 years (Table 1).
Fifty-one patients (98.4%) had diabetes mellitus (DM) of which 33 patients (63.4%) were found to have uncontrolled sugars (>250 mg/dL) and 15 patients (28.8%) had diabetic ketoacidosis (DKA). Renal involvement in the form of acute kidney injury was found in 14 patients (26.9%), while hepatic failure was seen in four patients (7.7%). Eight patients had systemic hypertension while three had cardiovascular disease. Cerebral involvement (ROC or stage III disease) was seen in eight patients (15.4%). Stage II disease was seen in 44 patients (84.6%). The mean duration of systemic symptoms was 13.12 ┬▒ 6.98 days whereas the mean duration of ocular symptoms was 7.28 ┬▒ 3.46 days.
Uncontrolled blood sugars at presentation (p = 0.008), presence of DKA (p = 0.03), blood urea levels at presentation (p = 0.034), and duration of ocular symptoms (blurring of vision, ptosis, proptosis, etc.) at presentation (p = 0.034) were all associated with poor treatment outcome on univariate analysis (Table 1). However, we could not find any significant association of the treatment outcomes with either the duration of systemic symptoms (fever, nasal discharge, headache, etc.) at presentation or at the stage of the disease (stage II or stage III).
Ocular assessment details at presentation are shown in Table 2. BCVA at presentation was 20 / 200 or better in 14 patients (26.9%), hand movements (HM) to 20 / 200 in 12 patients (23.1%), and no perception of light (NPL) in 22 patients (42.3%). One patient with bilateral involvement had NPL vision in both eyes. Vision could not be assessed in four patients due to altered sensorium. NPL vision at presentation was associated with poor treatment outcome (p = 0.001) on univariate analysis. Ptosis was noted in 40 patients (76.9%) and proptosis in 43 patients (82.7%) at presentation. Presence of ptosis was associated with poor outcome (p = 0.006) on univariate analysis. Relative afferent pupillary defect (RAPD) was noted in 29 patients (55.8%) and it was also associated with poor treatment outcome (p = 0.008) on univariate analysis. CRAO was seen in 16 patients (30.8%), cilioretinal artery occlusion in one patient, proliferative diabetic retinopathy (DR) in nine patients (17.3%), nonproliferative DR in 12 patients (23.1%). Presence of CRAO was associated with poor treatment outcome (p = 0.021). However, DR status was not associated with poor treatment outcome.
Conjunctival chemosis was noted in 49 patients (94.2%). Corneal involvement was found in 36 patients: neurotrophic keratitis in 11 patients (21.5%), while 25 patients (48.1%) had exposure keratitis. Restriction of extraocular movements was noted in 40 patients (76.9%). Of these clinical features, presence of conjunctival chemosis was noted to be associated with a poor treatment outcome (p = 0.01) along with ptosis (p = 0.03) on univariate analysis.
CECT was performed for all patients at or within 24 hours of presentation. Thirteen patients had only single sinus involvement, while 39 patients had two or more sinuses involved. Paranasal sinus involvement on CECT is shown in Table 3. Bony erosion with the contiguous invasion of the orbit was seen in 20 patients. Orbital apex involvement was seen in seven patients, cavernous sinus involvement in seven patients, and intracranial extension in six patients. Extraconal invasion and intraconal invasion were seen in 25 and 20 patients, respectively. Involvement of two or more sinuses (p = 0.024) and bony erosion of the orbital wall (p = 0.04) were associated with poor treatment outcome on univariate analysis (Table 3).
Diagnostic nasal endoscopy was performed in all the patients. Among them, 32 patients (61.0%) showed black-colored crusts in the nasal cavity. Samples taken during diagnostic nasal endoscopy were sent for microbiological evaluation. Potassium hydroxide mount showed either broad aseptate hyphae or fungal elements in 26 patients. Fungal culture details were available for 40 patients. Culture was positive for fungal growth in 18 patients: Mucor sporangiophores (two patients) and Rhizopus sporangiophores (16 patients) (Table 3).
Liposomal amphotericin B was initiated empirically in all 52 patients. Owing to the renal toxicity of the drug, renal adjustment of dosage was done in 14 patients. The full course of treatment was completed in 31 patients; 14 patients had expired before the completion of the course while seven patients were unable to afford the drug. Treatment regimen A (medical treatment plus nasal debridement) was given in 38 patients, regimen B (treatment A plus exenteration) in five patients, and regimen C (only medical treatment) in the remaining nine patients. Among these nine patients, four patients had cerebral involvement which was deemed unresectable, while five patients were unfit for surgery throughout their hospital stay. Nasal debridement alone through the transnasal endoscopic route (functional endoscopic sinus surgery, FESS) was performed in 26 patients. Partial maxillectomy was indicated in addition to local debridement in 12 patients. In three patients, maxillectomy was performed after FESS in a second sitting owing to inadequate clearance of the disease. Primary exenteration (regimen B) combined with debridement was performed in five patients. Neurosurgical drainage of abscess along with the transcranial injection of liposomal amphotericin B was given in three patients. Lateral canthotomy was needed in one patient to provide relief from orbital compartment syndrome. Final outcome was poorer as the treatment regimen progressed from A to C (p < 0.001). Surgical management for all the patients has been depicted in a flowchart in Fig. 1.
Overall, out of 52 patients, 19 patients (36.5%) had treatment success while 33 patients (63.5%) had treatment failure. Only 31 patients had completed full course of treatment while remaining 21 either expired (14 patients) or had taken discharge against medical advice (seven patients). All these seven patients had deterioration of their physical conditions during their stay in the hospital and were considered as treatment failure. Patient survival was, therefore, considered as 31 of 52 (59.6%). Out of the 31 patients who had survived, clinical improvement was seen in 19 patients, while five patients required exenteration and the conditions remained unchanged in seven patients. BCVA at last follow-up in 31 patients completing the full course of treatment was 20 / 200 or better in 14 patients, HM to 20 / 200 in six patients, and NPL in 11 patients. Vision survival (vision of HM or better) was, therefore, noted in 20 of 52 patients (38.5%) and globe survival was noted in 26 of 52 patients (52.0%). Improvement in the extraocular movements and ptosis was observed in 10 of 31 patients (32.3%). Resolution of RAPD was observed in five of 31 patients (16.1%). Treatment outcome for all 52 patients has been shown in Fig. 2 and Table 4.
On univariate analysis, factors associated with poor treatment outcome were uncontrolled blood sugar (>250 mg/dL), DKA, random blood sugar at presentation, blood urea, duration of ocular symptoms, NPL vision at presentation, ptosis, conjunctival chemosis, RAPD, CRAO, two or more sinus involvement, bony erosion of orbital wall, etc. (Tables 1-3). Multicollinearity test was performed to detect any possible intercorrelations between the various independent variables that were found significant on univariate analysis. However, none of the factors were found to be intercorrelated. Logistic regression analysis was subsequently performed, and adjusted odds ratio (aOR) was calculated for each factor. Factors found to have significant associations with poor treatment outcome on logistic regression analysis were uncontrolled blood sugar (aOR, 1.17; p = 0.018), NPL at presentation (aOR, 10.67; p = 0.006), ptosis at presentation (aOR, 3.90; p = 0.03), conjunctival chemosis (aOR, 7.11; p = 0.024), RAPD (aOR, 10.60; p = 0.01), CRAO at presentation (aOR, 3.54; p = 0.021), and two or more sinus involvement (aOR, 4.90; p = 0.009) (Table 4).
DM is the most common risk factor for mucormycosis accounting for 60% to 80% of the reported cases of mucormycosis worldwide. Other predisposing conditions include DKA, immunosuppressive therapy, chronic renal failure, cirrhosis, hematological malignancies, solid organ transplantation, and HIV infection [11,12]. In our study, DM was seen in 51 of 52 patients (98.1%), DKA in 15 of 52 patients (28.8%), renal involvement in 14 of 62 patients (26.9%), and hepatic failure in four of 52 patients (7.7%). However, none of the patients that were diagnosed and treated at our center as orbital mucormycosis during the current study period had hematological malignancy or organ transplantation as risk factors. This is unlike few other studies where these risk factors were noted as frequently as DM [6,13]. Similar to this study, Kashkouli et al. [14] had reported type 1 DM, type 2 DM, and renal failure in 10 (15.9%), 33 (52.4%), and two of 63 cases (3.2%), respectively. In another study by Yohai et al. [15] DM, DKA, and renal disease was present in 87 (60%), 43 (30%), and 21 of 145 cases (14%), respectively. Uncontrolled blood sugars at presentation, in fact, was a factor associated with poor treatment outcome on both univariate analysis and logistic regression analysis in this study. Control of blood sugar levels is, therefore, very essential for successful treatment of a case of orbital mucormycosis.
The signs and symptoms of ROCM depend upon the route of entry and the involved structures. Initial presentation may be in the form of loss of vision or nerve palsy causing restriction of extraocular movements and diplopia followed by ptosis, proptosis, internal ophthalmoplegia, and CRAO. Intracranial spread may cause severe headache, altered sensorium, and finally death [11,16-18]. Ocular findings seen in this study were conjunctival chemosis (94.2%), proptosis (82.7%), ptosis (76.9%), restriction of extraocular movements (76.9%), RAPD (55.8%), exposure keratitis (48.1%), NPL vision at presentation (42.3%), neurotrophic keratitis (21.5%), CRAO (30.8%), proliferative DR (17.3%), and nonproliferative DR (23.1%). In the study by Bhansali et al. [19], where 35 cases were studied, the ocular features noted were ophthalmoplegia (89%), proptosis (83%), loss of vision (80%), chemosis (74%), periorbital swelling (66%), CRAO (20%), etc. Kashkouli et al. [14] studied 63 cases, and the reported ocular features include proptosis (39.2%), extraocular movement abnormality (83.5%), frozen eye (55.7%), loss of vision (24.1 %), etc. The duration of ocular symptoms at initial presentation also served as a predictor for poor treatment outcome on univariate analysis in our study. Delay in diagnosis and treatment was noted to be associated with poor patient survival in prior studies as well [15,19].
Presence of ptosis, RAPD, CRAO, NPL vision at presentation, and conjunctival chemosis were all significantly associated with poor treatment outcome on univariate analysis. On logistic regression analysis, NPL vision at presentation (aOR, 10.67), conjunctival chemosis (aOR, 7.11), and RAPD (aOR, 10.60) maintained significant association with poor treatment outcome. Presence of these ocular features at presentation may serve as predictors of poor treatment outcome. The current study, therefore, identifies these ocular features as predictors that may herald worsening of the systemic disease and, thereby, determine the need for more aggressive treatment in such cases. In the study by Kashkouli et al. [14], frozen eye (OR, 4.6) was associated with poor outcome on multivariate analysis. In the study by Bhansali et al. [19], presence of eyelid gangrene (p < 0.05) was related to poor survival.
CECT and magnetic resonance imaging are both invaluable tools for diagnosing and assessing the disease progression in ROCM [20]. Maxillary sinus (84.61%) followed by ethmoidal sinus (75%) were the two most affected sinuses in our study populationŌĆöfindings similar to the study by Abdollahi et al. [21]. In the study by Kashkouli et al. [14], ethmoidal sinus (74.6%) was most commonly affected followed by the maxillary sinus (68.3%). Two or more sinuses were involved in 63.3% of cases in their study but this was not associated with poor treatment outcome. In contrast, 39 patients (75%) in our study had two or more sinuses involved on radiological imaging and this was a significant factor for poor treatment outcome in both univariate analysis (OR, 4.22) and logistic regression analysis (aOR, 4.90). Radiological findings can, therefore, serve as predictors of severe disease and guide the treatment course.
Vision survival was noted in 38.5% of patients and globe survival in 52.0% of patients in our study. Vision loss in ROCM can result from either CRAO, exenteration, or cavernous sinus thrombosis [3-5]. In our study, 16 patients had CRAO, and seven patients had cavernous sinus thrombosis. CRAO was associated with poor treatment outcome on univariate analysis which, however, lost significance on logistic regression analysis. Indications of orbital exenteration include progressive disease with proptosis, cranial nerve involvement, CRAO, etc. [5,14,22-24]. Exenteration can improve patient survival in cases with intracranial extension and rapid progression. However, survival may not improve if the patients already have aggressive disease prior to exenteration [25,26]. In this study, five patients needed exenteration and globe survival was noted in 26 of total 31 patients that completed full course of medical treatment. We could not analyze the factors associated with globe survival or vision survival in our study. In the study by Kashkouli et al. [14], higher white blood cell count was the only factor that was found to be associated with the need for exenteration. No other studies have evaluated the factors affecting vision and globe survival. Future prospective studies may, therefore, be planned to determine the factors that can affect the vision and globe survival in ROCM. Our current study showed a patient survival rate of 59.6%, which is comparable to the survival ratesŌĆö73.3%, 70%, 57%ŌĆöof the studies done by Abdollahi et al. [21], Roden et al. [6], and Kashkouli et al. [14], respectively. However, one important aspect to be considered here is that the early stage I of the disease was not included in our study which has affected the survival rate in this study. More advanced diseases, stage II and stage III with poorer survival rates, were included in the study. Prompt diagnosis, early initiation of treatment with systemic antifungals and surgical debridement of the sinuses are the key steps to a higher survival rate in ROCM.
Strength of our study lies in its large sample size. Only few prior studies had equivalent or more sample size [14,15]. Also, the current study analyzed and identified ocular and radiological features as predictors of aggressive systemic disease and poor treatment outcome. One limitation of the study is the retrospective study design. However, ROCM being a rare and aggressive disease, patient recruitment and follow-up may be difficult for a prospective study design.
Patient survival, globe survival, and vision survival were 59.6%, 52.0%, and 38.5%, respectively in this study. DM was the most common underlying systemic disease. High blood sugar at presentation, NPL vision at presentation, ptosis, conjunctival chemosis, RAPD, bony erosion of orbital wall, and two or more sinus involvement were the major predictors for poor treatment outcome.
References
1. Wali U, Balkhair A, Al-Mujaini A. Cerebro-rhino orbital mucormycosis: an update. J Infect Public Health 2012;5:116-26.


3. Sharma RR, Pawar SJ, Delmendo A, et al. Fatal rhino-orbito-cerebral mucormycosis in an apparently normal host: case report and literature review. J Clin Neurosci 2001;8:583-6.


4. Peterson KL, Wang M, Canalis RF, Abemayor E. Rhinocerebral mucormycosis: evolution of the disease and treatment options. Laryngoscope 1997;107:855-62.


5. Onerci M, Gursel B, Hosal S, et al. Rhinocerebral mucormycosis with extension to the cavernous sinus: a case report. Rhinology 1991;29:321-4.

6. Roden MM, Zaoutis TE, Buchanan WL, et al. Epidemiology and outcome of zygomycosis: a review of 929 reported cases. Clin Infect Dis 2005;41:634-53.


7. Andreani G, Fadda G, Gned D, et al. Rhino-orbital-cerebral mucormycosis after allogeneic hematopoietic stem cell transplantation and isavuconazole therapeutic drug monitoring during intestinal graft versus host disease. Mediterr J Hematol Infect Dis 2019;11:e2019061.




8. Sun HY, Forrest G, Gupta KL, et al. Rhino-orbital-cerebral zygomycosis in solid organ transplant recipients. Transplantation 2010;90:85-92.


10. Nithyanandam S, Jacob MS, Battu RR, et al. Rhino-orbito-cerebral mucormycosis: a retrospective analysis of clinical features and treatment outcomes. Indian J Ophthalmol 2003;51:231-6.

11. Mukherjee B, Raichura ND, Alam MS. Fungal infections of the orbit. Indian J Ophthalmol 2016;64:337-45.



12. Anders UM, Taylor EJ, Martel JR, Martel JB. Acute orbital apex syndrome and rhino-orbito-cerebral mucormycosis. Int Med Case Rep J 2015;8:93-6.


13. Lanternier F, Dannaoui E, Morizot G, et al. A global analysis of mucormycosis in France: the RetroZygo Study 2005-2007. Clin Infect Dis 2012;54:Suppl 1. S35-43.


14. Kashkouli MB, Abdolalizadeh P, Oghazian M, et al. Outcomes and factors affecting them in patients with rhino-orbito-cerebral mucormycosis. Br J Ophthalmol 2019;103:1460-5.


15. Yohai RA, Bullock JD, Aziz AA, Markert RJ. Survival factors in rhino-orbital-cerebral mucormycosis. Surv Ophthalmol 1994;39:3-22.


16. Jiang N, Zhao G, Yang S, et al. A retrospective analysis of eleven cases of invasive rhino-orbito-cerebral mucormycosis presented with orbital apex syndrome initially. BMC Ophthalmol 2016;16:10.




17. Shah K, Dave V, Bradoo R, et al. Orbital exenteration in rhino-orbito-cerebral mucormycosis: a prospective analytical study with scoring system. Indian J Otolaryngol Head Neck Surg 2019;71:259-65.




18. Luo QL, Orcutt JC, Seifter LS. Orbital mucormycosis with retinal and ciliary artery occlusions. Br J Ophthalmol 1989;73:680-3.



19. Bhansali A, Bhadada S, Sharma A, et al. Presentation and outcome of rhino-orbital-cerebral mucormycosis in patients with diabetes. Postgrad Med J 2004;80:670-4.



20. Som PM, Dillon WP, Curtin HD, et al. Hypointense paranasal sinus foci: differential diagnosis with MR imaging and relation to CT findings. Radiology 1990;176:777-81.


21. Abdollahi A, Shokohi T, Amirrajab N, et al. Clinical features, diagnosis, and outcomes of rhino-orbito-cerebral mucormycosis: a retrospective analysis. Curr Med Mycol 2016;2:15-23.
22. Kursun E, Turunc T, Demiroglu YZ, et al. Evaluation of 28 cases of mucormycosis. Mycoses 2015;58:82-7.


23. Dhiwakar M, Thakar A, Bahadur S. Improving outcomes in rhinocerebral mucormycosis: early diagnostic pointers and prognostic factors. J Laryngol Otol 2003;117:861-5.


24. Ketenci I, Unlu Y, Kaya H, et al. Rhinocerebral mucormycosis: experience in 14 patients. J Laryngol Otol 2011;125:e3.


Fig.┬Ā1
Flowchart showing surgical management of the cases. FESS = functional endoscopic sinus surgery.
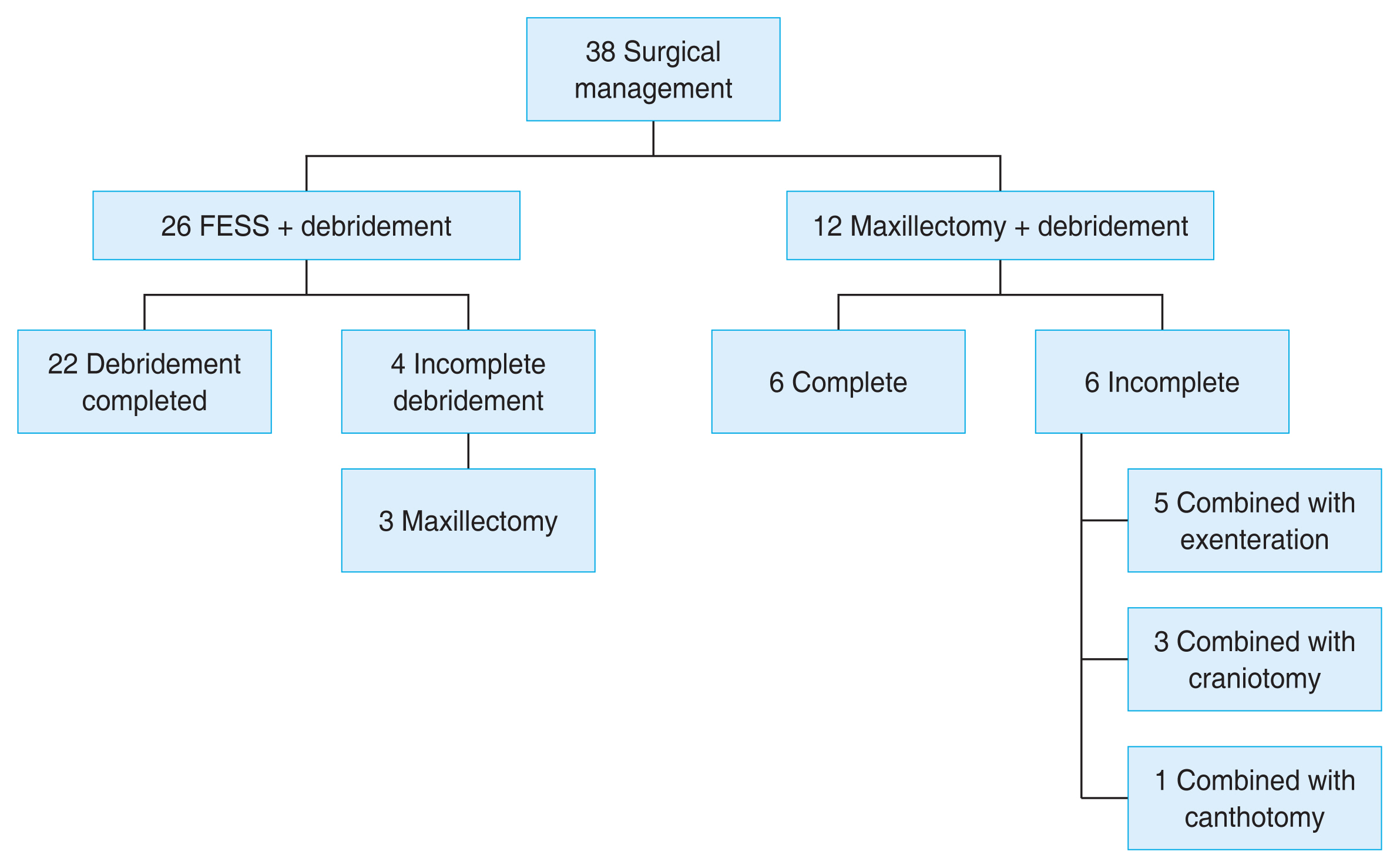
Fig.┬Ā2
Flowchart showing treatment outcomes. L-AmpB = liposomal amphotericin B; Amp = amphotericin.

Table┬Ā1
Baseline sociodemographic and clinical characteristics of all the patients
| Characteristic | Total (n = 52) | Treatment success (n = 19) | Treatment failure (n = 33) | p-value |
|---|---|---|---|---|
| Sex | ||||
| ŌĆāMale | 36 (69.2) | 14 (73.7) | 22 (66.7) | 0.758 |
| ŌĆāFemale | 16 (30.7) | 5 (26.3) | 11 (33.3) | |
| Age (yr) | 51.84 ┬▒ 14.06 (8-74) | - | - | 0.700 |
| Associated systemic disease and laboratory characteristic | ||||
| ŌĆāDiabetes mellitus | 51 (98.1) | 19 (100) | 32 (97.0) | |
| ŌĆāUncontrolled blood sugar (>250 mg/dL) | 33 (63.4) | 7 (36.8) | 26 (78.8) | 0.008* |
| ŌĆāDiabetic ketoacidosis | 15 (28.8) | 3 (15.8) | 12 (36.4) | 0.030* |
| ŌĆāRandom blood sugar at presentation | 266.85 ┬▒ 129.02 | 19 (100) | 33 (100) | 0.041* |
| ŌĆāUrea | 48.19 ┬▒ 29.40 | 19 (100) | 33 (100) | 0.034* |
| ŌĆāCreatinine | 1.41 ┬▒ 1.13 | 19 (100) | 33 (100) | 0.580 |
| ŌĆāHypertension | 8 (15.4) | 3 (15.8) | 5 (15.2) | 0.841 |
| ŌĆāRenal failure (e-GFR) | 14 (26.9) | 4 (21.1) | 10 (30.3) | 0.043* |
| ŌĆāHepatic failure | 4 (7.7) | 2 (10.5) | 2 (6.1) | 0.980 |
| Duration of systemic symptomŌĆĀ (day) | 13.12 ┬▒ 6.98 (2-45) | 19 (100) | 33 (100) | 0.312 |
| Duration of ocular symptomŌĆĪ (day) | 7.28 ┬▒ 3.46 (1-16) | 19 (100) | 33 (100) | 0.034* |
| Stage of disease | ||||
| ŌĆāStage II (rhino-orbital) | 44 (84.6) | 15 (78.9) | 29 (87.9) | 0.443 |
| ŌĆāStage III (rhino-orbito-cerebral) | 8 (15.4) | 4 (21.1) | 4 (45.5) |
Table┬Ā2
Ocular features at presentation
| Variable | Total | Treatment success | Treatment failure | p-value |
|---|---|---|---|---|
| BCVA at presentation (n = 48) | - | |||
| ŌĆā>20 / 200 | 14 | 5 | 9 | |
| ŌĆā20 / 200 (HMCF) | 12 | 6 | 6 | |
| ŌĆāNPL | 22 | 5 | 17 | |
| Signs and symptoms | ||||
| ŌĆāNPL vision at presentation | 22 | 5 | 17 | 0.001* |
| ŌĆāProptosis | 43 | 15 | 28 | 0.588 |
| ŌĆāPtosis | 40 | 10 | 30 | 0.006* |
| ŌĆāConjunctival chemosis | 49 | 8 | 41 | 0.005* |
| ŌĆāRestriction of EOM | 40 | 12 | 28 | 0.037* |
| ŌĆāRelative afferent pupillary defect | 29 | 7 | 22 | 0.008* |
| ŌĆāNeurotrophic keratitis | 11 | 3 | 8 | 0.726 |
| ŌĆāExposure keratitis | 25 | 12 | 13 | 0.970 |
| ŌĆāCentral retinal artery occlusion | 16 | 2 | 14 | 0.016* |
| ŌĆāCilioretinal artery occlusion | 1 | 0 | 1 | - |
| ŌĆāChoroidal folds | 1 | 0 | 1 | - |
| ŌĆāProliferative DR | 9 | 1 | 8 | 0.074 |
| ŌĆāNonproliferative DR | 12 | 4 | 8 | 0.583 |
| ŌĆāAny DR stage | 21 | 5 | 16 | 0.118 |
Table┬Ā3
Imaging findings and microbiological profile
| Variable | Total | Treatment success | Treatment failure | p-value |
|---|---|---|---|---|
| Sinus and orbital involvement | ||||
| ŌĆāMaxillary sinus | 44 | 13 | 31 | - |
| ŌĆāEthmoidal sinus | 39 | 13 | 26 | - |
| ŌĆāSphenoidal sinus | 25 | 6 | 19 | - |
| ŌĆāFrontal sinus | 16 | 4 | 12 | - |
| ŌĆāOne sinus involved | 13 | 9 | 4 | 0.024* |
| ŌĆāTwo or more sinuses involved | 39 | 10 | 29 | - |
| ŌĆāBony erosion of orbital wall | 20 | 5 | 15 | 0.040* |
| ŌĆāNoncontiguous orbital involvement | 37 | 11 | 26 | 0.110 |
| ŌĆāExtraconal invasion | 25 | 5 | 20 | 0.039* |
| ŌĆāIntraconal invasion | 20 | 6 | 14 | 0.430 |
| ŌĆāOrbital apex involvement | 7 | 1 | 6 | 0.242 |
| ŌĆāIntracranial extension | 6 | 3 | 3 | 0.656 |
| ŌĆāCavernous sinus thrombosis | 7 | 1 | 6 | 0.242 |
| Microbiological culture profile | 40 (100) | 18 (45.0) | 22 (55.0) | - |
| ŌĆāNegative culture | 22 (55.0) | 9 (50.0) | 13 (59.1) | |
| ŌĆāMucor | 2 (5.0) | 1 (5.6) | 1 (4.5) | |
| ŌĆāRhizopus | 16 (40.0) | 8 (44.4) | 8 (36.4) | |
Table┬Ā4
Logistic regression analysis to identify factors affecting treatment outcome
- TOOLS



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




