Spontaneous Recovery of Traumatic Unilateral Superior Oblique Palsy and Ocular Factors for Predicting Prognosis
Article information
Abstract
Purpose
This study evaluated the prognosis of patients with traumatic unilateral superior oblique palsy (SOP) and clinical factors associated with spontaneous resolution.
Methods
Medical records of patients with traumatic unilateral SOP who visited two hospitals (Yeungnam University Hospital and Daegu Catholic University Medical Center) between January 2015 and June 2020 were reviewed retrospectively. When traumatic unilateral SOP did not recover within at least 1 year of follow-up, no spontaneous resolution was considered. Both traumatic and ocular factors were evaluated to evaluate their association with spontaneous recovery.
Results
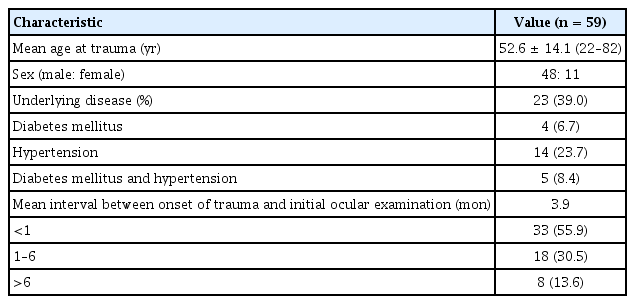
Fifty-nine patients (mean age, 52.6 years; 48 male patients) were enrolled in this study. The mean interval from trauma to initial presentation was 3.9 months. The mean vertical deviation at initial presentation was 6.34 ± 5.22 prism diopters (PD) (range, 0–25 PD). During the mean 24.1-month follow-up period, 28 patients (47.5%) achieved spontaneous resolution of SOP. Thirteen patients underwent surgical treatment for SOP. Vertical deviation <6 PD at the initial visit and low fundus torsion in the nonparetic eye and both eyes were significantly associated with spontaneous recovery (p < 0.05, logistic regression analysis). Traumatic factors, including the trauma type, presence of intracranial lesion, loss of consciousness, and Glasgow Coma Scale score, were not associated with spontaneous recovery.
Conclusions
In this multicenter study, spontaneously recovery was achieved in 47.5% patients with traumatic unilateral SOP. Even mild head trauma can lead to permanent SOP. Ocular factors, including the angle of deviation and fundus torsion, may better predict spontaneous resolution than traumatic factors in patients with traumatic unilateral SOP.
Cranial nerve palsy and binocular diplopia can be caused by head trauma [1–5]. Even mild head trauma with an initial Glasgow Coma Scale (GCS) score of 14 to 15 after impact can lead to cranial nerve palsy [6]. Among the cranial nerves related to ocular motility, palsy of the fourth cranial nerve, known as superior oblique palsy (SOP), is most commonly caused by trauma [7,8]. The fibers of the trochlear nerve, which supplies the superior oblique muscles, decussate and exit the brainstem on its dorsal surface. Patients with traumatic SOP should be observed for several months before surgical realignment is considered as partial or complete recovery occurs spontaneously in many cases.
Recently, the types of trauma have become more complex and severe [3,9,10]. Hence, we believe that the prognosis of traumatic SOP has diversified and changed. Moreover, most previous studies have evaluated traumatic factors in patients with traumatic SOP and investigated the association between these factors and traumatic SOP [1,2,6]. However, the association between ocular factors, evaluated by clinical examination, and spontaneous recovery of SOP has rarely been evaluated. Therefore, we evaluated the prognosis of patients with traumatic unilateral SOP and clinical factors that were associated with spontaneous recovery.
Materials and Methods
Ethics statement
The present study adhered to the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board of Yeungnam University Hospital. Given the retrospective nature of the study and the use of anonymized patient data, requirements for informed consent were waived by the institutional review board.
Patient selection
Medical records of patients with binocular diplopia after trauma who consulted the eye clinic of two hospitals (Yeungnam University Hospital and Daegu Catholic University Medical Center) on outpatient or inpatient basis and those who presented to the emergency department these hospitals between January 2015 and June 2020 were reviewed retrospectively. Patients who were diagnosed with traumatic unilateral SOP based on clinical evaluations were included in the study. Patients with a history of other strabismus, a presence of diplopia before the trauma, any orbital wall fracture, concomitant oculomotor or abducens nerve palsies, and any signs of bilateral SOP were excluded.
Patient evaluation
Data on the type and onset of trauma were collected using the patient’s self-reported medical history or previous medical records from the emergency department. The presence of loss of consciousness and GCS score at trauma were evaluated using prior medical records when possible. The presence of intracranial traumatic lesions was identified using previous brain images. The definition of mild head trauma reported by Li et al. [6] was used: (1) immediate or delayed occurrence of ocular movement nerve palsy after recent head injury, (2) GCS score of 14 to 15 after impact regardless of the history of transient consciousness disturbance, (3) no definite skull fracture or intracranial traumatic lesions (hemorrhage, cerebral contusion, or laceration) seen on computed tomography or magnetic resonance imaging, and (4) no other definite cause responsible for the genesis of ocular movement nerve palsy identified by radiological, laboratory, or clinical investigations.
At the initial visit to the eye department, all patients underwent full ophthalmologic examination, including assessment of visual acuity and ocular alignment status, slit-lamp biomicroscopy, and fundus examination. The angle of deviation was measured using the alternate prism cover test at 6-m and 33-cm fixation. All the included patients had undergone the Bielschowsky head tilt test. Fundus photographs were taken to evaluate fundus torsion. The Kowa fundus camera (VK-2; Kowa Optimed Inc., Tokyo, Japan) and Optos Carfornia (Optos, Dunfermline, UK) were used to acquire fundus photographs. Before imaging, all patients were instructed to fixate on the camera’s internal fixation marker with their head in the neutral position on the chin rest and head support by the expert technician. Unwanted head tilting was prevented by aligning the patient’s lateral canthi to side marks on the fundus camera. Torsional deviation, which is the angle between a horizontal line passing through the optic disc center and a line connecting the fovea and optic disc center, was measured using Image J software ver. 1.53e (National Institute of Health, Bethesda, MD, USA) [11,12]. In addition, because extorsion may appear in the nonparetic eye, the sum of torsional angles in both eyes was evaluated to analyze changes in extorsion [11,12].
Prism glasses or a monocular patch were prescribed to relieve binocular diplopia. Complete recovery was defined as cessation of subjective diplopia accompanied by an improvement in ocular deviation. When traumatic SOP did not spontaneously recover within at least 1 year of follow-up, no spontaneous recovery was considered. Surgical intervention was considered in cases of no spontaneous recovery after at least 1 year of follow-up.
Statistical analysis
Continuous data are expressed as mean and standard deviation, and categorical data are expressed as counts and percentages. Data on patients’ basic characteristics, traumatic factors, and ocular factors were collected. Logistic regression analysis was performed to evaluate factors associated with spontaneous recovery of SOP. Data were analyzed using IBM SPSS ver. 20.0 (IBM Corp., Armonk, NY, USA). A p-value of <0.05 was considered statistically significant.
Results
Basic characteristics of the included patients
Fifty-nine patients (mean age, 52.6 years; 48 male patients, 11 female patients) were enrolled. The basic characteristics of the included patients are shown in Table 1. The mean interval from trauma to initial presentation was 3.9 months (range, 0–43 months). In total, 33 patients (55.9%) visited or consulted the eye department for binocular diplopia within 1 month of trauma. However, eight patients (13.6%) first presented to the clinic after >6 months of trauma.
Traumatic and ocular factors
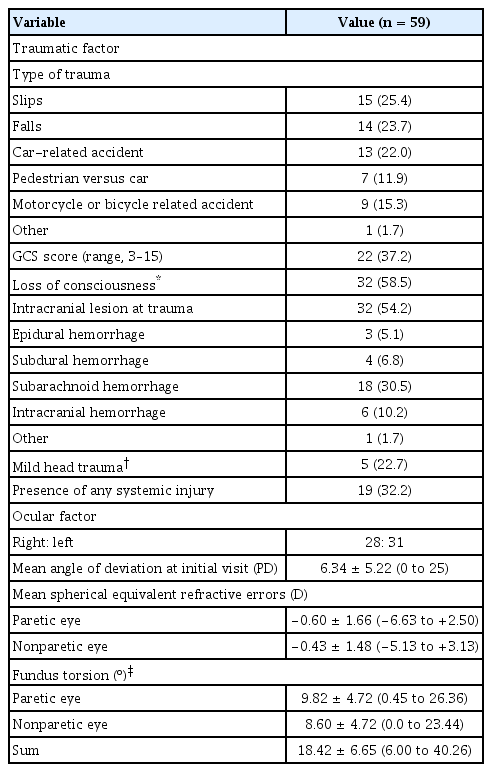
The traumatic and ocular factors are listed in Table 2. Falls and car-related accidents were the most common types of trauma. The GCS score was assessed in 22 patients. In total, 32 patients (54.2%) had an intracranial lesion on imaging investigations performed at the onset of trauma. Among these patients, five patients met the definition of mild head trauma. Nineteen patients (32.2%) had systemic injuries, including injuries to the chest and the abdomen, and fractures of an extremity. All patients showed similar laterality of the affected eye. The mean vertical deviation at the initial visit was 6.34 prism diopters (PD) (range, 0–25 PD). The mean degree of fundus extorsion was 9.82º and 8.60º in the paretic and nonparetic eye, respectively. Thirty-five patients (60.3%) exhibited extorsion of the paretic eye, and 20 patients (34.5%) exhibited extorsion of the nonparetic eye.
Prognostic factors for spontaneous recovery of traumatic unilateral SOP
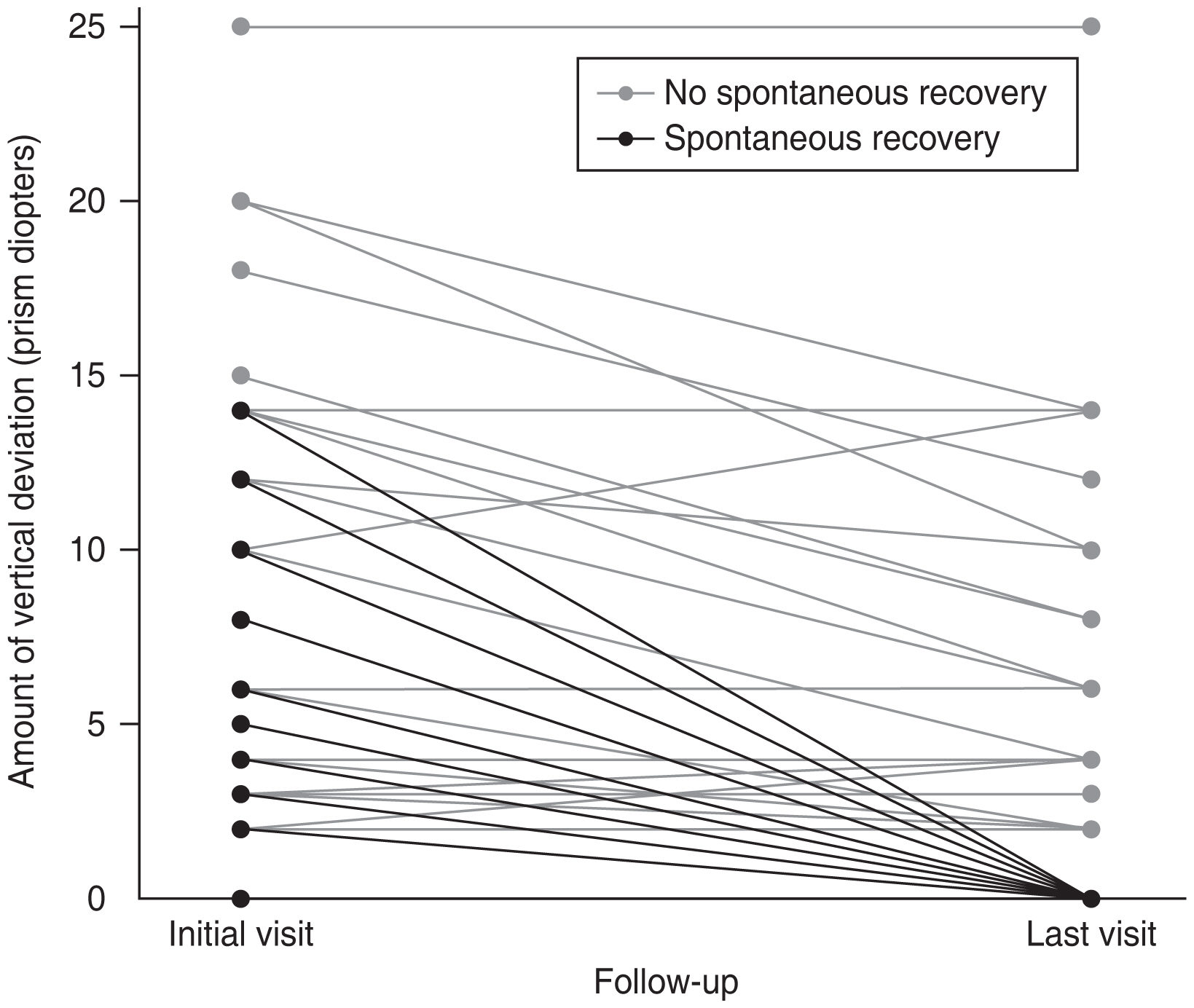
During the mean 24.1-month follow-up period after the initial visit, 28 patients (47.5%) achieved spontaneous recovery of traumatic unilateral SOP (Fig. 1). Four of the five patients with mild head trauma also achieved spontaneously recovery of traumatic unilateral SOP. Of the 31 patients who did not achieve spontaneous recovery, 13 patients (41.9%) underwent surgical treatment for SOP. The basic characteristics, traumatic factors, and ocular factors were evaluated to identify factors associated with spontaneous recovery of SOP. The mean age at trauma, sex, the presence of underlying disease, and the interval between onset of trauma and ocular examination were not significantly associated with spontaneous recovery (all p > 0.05). Traumatic factors, including the type of trauma, the presence of intracranial lesions, loss of consciousness, and the GCS score, were not associated with spontaneous recovery. Vertical deviation <6 PD at the initial visit was significantly associated with spontaneous recovery (p = 0.025, logistic regression analysis). High fundus torsion in the paretic eye was associated with less spontaneous recovery, but it was not statistically significant (p = 0.154). Increased fundus torsion in the nonparetic eye and the sum of torsion in both eyes were significantly associated with less spontaneous resolution (p = 0.021 and p = 0.010, respectively; logistic regression analysis).
Discussion
In this multicenter study, spontaneous recovery was observed in 47.5% patients with traumatic unilateral SOP. Relatively mild head trauma can occur traumatic SOP, leading to permanent SOP. Ocular factors, including the angle of deviation and amount of fundus torsion, may be better predictive factors than traumatic factors for spontaneous recovery.
Direct injury due to stretching and/or compressive forces or impairment of the vascular supply of the nerve may be reasonable explanations for SOP after trauma. Traumatic SOP and binocular diplopia are not fatal; however, they are troublesome for the affected patients. Sharma et al. [4] evaluated ocular manifestations of head injury and reported that the third and fourth cranial nerves were the most commonly involved cranial nerves. Ocular motor nerve palsies are more frequently observed in frontal and parietal injuries. They suggested that both ophthalmological and neurosurgical approaches are important in managing patients with head injury for good visual outcomes and better prognosis. However, detailed ocular examinations may not be possible in the acute period in cases of severe trauma. Ocular examination should be performed in patients with head trauma, especially those with visual disturbance.
In this study, 28 patients (47.5%) achieved spontaneous recovery of traumatic unilateral SOP during the mean follow-up of 24.1 months. The recovery rate for traumatic SOP has decreased in recent years compared with that reported previously; 61.9% of the patients (13 of 21 patients) achieved spontaneous recovery in a study by Sydnor et al. [7] in 1982, while 23.1% of the patients (nine of 39 patients) achieved spontaneous recovery in a study by Mollan et al. [8] in 2008. This may be attributed to the more severe complexity and extent of trauma in recent years. The prevalence and severity of head and ocular trauma have changed over time [10].
The prognostic factors for traumatic SOP are controversial. Most previous studies have focused on traumatic and ocular factors based on clinical evaluation [1,6]. Therefore, we investigated both traumatic and ocular factors in patients with traumatic SOP and evaluated its association with spontaneous recovery. In this study, the traumatic factors were not significantly associated with spontaneous recovery. A previous study by Li et al. [6] reported that the imaging results after trauma can be used as a prognostic factor for spontaneous recovery. However, interpretation of results may vary according to the clinician. They also pointed out that mild head trauma does not mean mild injury or favorable recovery of ocular movement nerve palsy after mild head trauma [6]. These results are consistent with our results. In our study, a 66-year-old male patient with hypertension developed permanent SOP after mild head trauma (Fig. 2A, 2B). He had diplopia during the 2-year follow-up period. Although he had mild head trauma, it is possible that the large vertical angle of 10 PD and a sum fundus torsion angle of 25° for both eyes at the initial visit had a negative effect on recovery from traumatic unilateral SOP. Therefore, traumatic factors alone may have limited value in predicting spontaneous recovery.
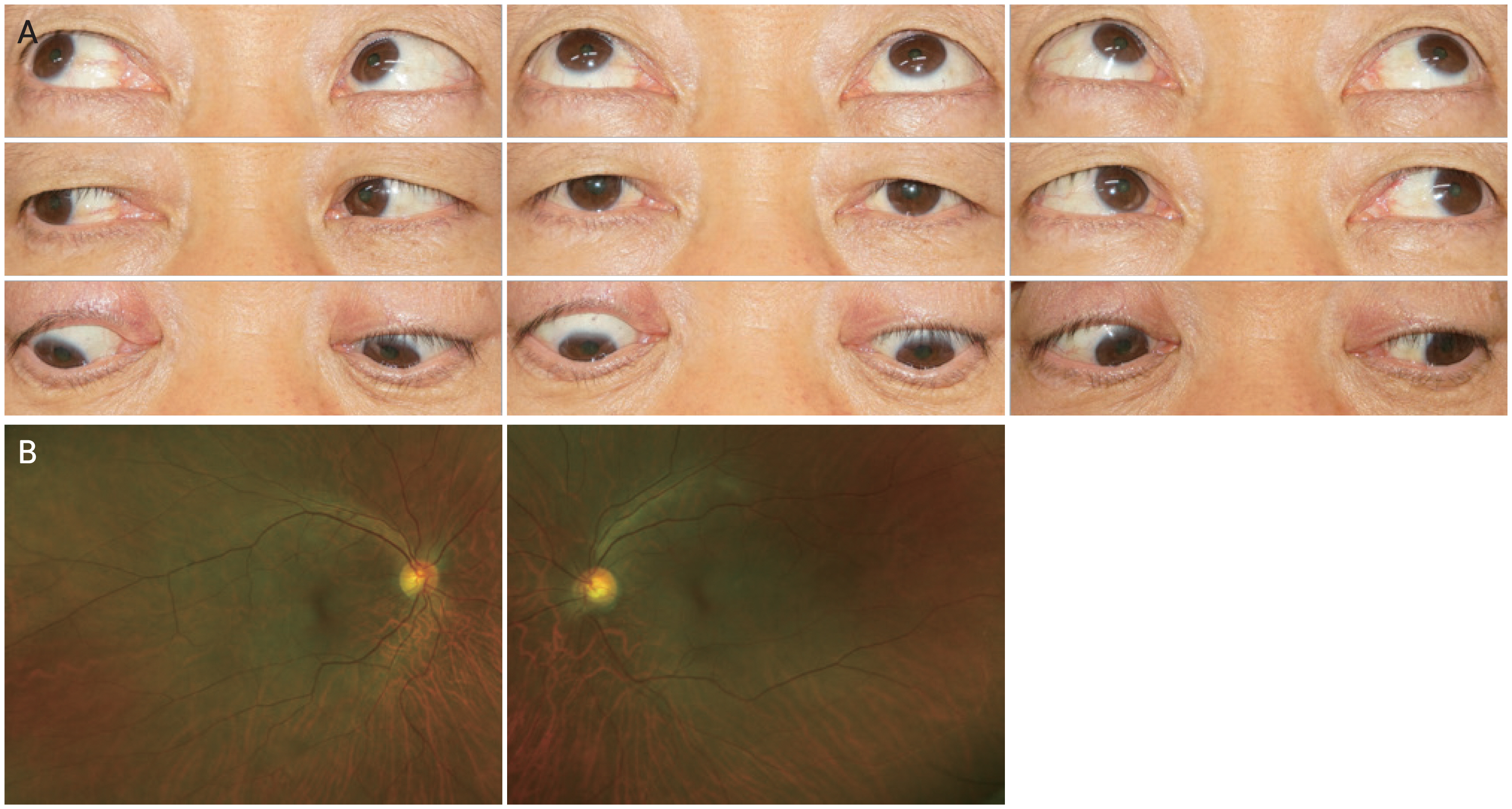
Images of (A) nine diagnostic positions of gaze and (B) fundus of both eyes. A 66-year-old male patient developed permanent superior oblique palsy after mild head trauma. He showed the hypertropia and elevation in adduction of the right eye. The fundus photo of right eye showed the extorsion. Informed consent for publication of the clinical images was obtained from the patient.
Ocular factors, including the amount of vertical deviation and fundus torsion, were significantly associated with spontaneous recovery. Vertical deviation <6 PD at the initial visit was significantly associated with the spontaneous resolution of traumatic SOP. These results are similar to those of a previous study by Oh and Oh [13] who analyzed the clinical outcomes and etiology of SOP associated with acute vertical diplopia. This study showed that the initial deviation angle was different between the recovery and nonrecovery groups. A larger amount of vertical deviation may be associated with more severe neurologic trauma and may prevent spontaneous recovery.
Kim and Lim [14] compared ocular torsion between patients with congenital and acquired unilateral SOP. In this study, patients with SOP had variable fundus torsion of varying etiologies. They suggested that unilateral SOP might initially induce paretic ocular extorsion and then induce ocular dominance, cyclofusion, or a neural adaptation mechanism in the nonparetic eye, increasing the total amount of extorsion in both eyes [14,15]. We suspected that more severe nerve damage leads had a greater effect on compensatory ocular torsion mechanisms and results in more fundus extorsion in the nonparetic eye. Hence, we suggest that the fundus findings in both paretic and non-paretic eyes should be checked to predict prognosis, even in patients with unilateral SOP.
This retrospective study has several limitations. First, the interval between trauma and initial ocular examination differed according to the patient’s clinical course. This is because patients with severe systemic injury may require more time to undergo detailed ocular examination. Future studies will include patients with the same interval between trauma and ocular examination. Second, patients with bilateral SOP were excluded from the study because of the small number of these patients. Bilateral SOP can occur after trauma based on its anatomical characteristics. A previous study showed that spontaneous recovery occurred less frequently in patients with bilateral SOP than in those with unilateral SOP [7]. Further studies including both patients with unilateral and bilateral SOP are needed to improve our understanding of the clinical course of traumatic SOP.
In conclusion, spontaneously recovery was achieved in 47.5% of the patients with traumatic unilateral SOP. Even mild head trauma can lead to permanent SOP. Ocular factors, including the angle of deviation and fundus torsion, may better predict spontaneous resolution than traumatic factors in patients with traumatic unilateral SOP. Therefore, ocular examination should be performed in patients with head trauma, especially those with visual disturbance.
Acknowledgements
None.
Notes
Conflicts of Interest: None.
Funding: None.