Persistent epithelial defects (PED) result from the failure of rapid re-epithelialization and closure even with standard supportive treatment [
1-
3]. The corneal epithelial healing process can be disrupted from defective epithelial adhesion, limbal stem cell deficiency, neurotrophic keratitis, mechanical injuries including exposure keratopathy, and inflammatory conditions such as keratoconjunctivitis sicca, rosacea, infectious keratitis, peripheral ulcerative keratitis, and autoimmune diseases [
1]. Regardless of the cause, when damaged corneal epithelium is not managed properly, it can progress to corneal ulcer, conjunctivalization, and neovascularization, ultimately resulting in corneal opacity and permanent visual loss [
2,
3]. Even with prompt diagnosis and supportive medical treatment, the visual prognosis for PED is poor due to slow recovery [
4,
5]. Therefore, additional surgical treatments such as tarsorrhaphy, conjunctival flap, and amniotic membrane transplantation (AMT) may be considered for improved results with regard to promoting regeneration of corneal epithelium in PED from various corneal diseases [
4].
AMT has been used in various corneal diseases since its first usage in ophthalmology by de Rotth [
6] in 1940 for conjunctival loss repair; evidence of its efficacy in treating corneal ulcers has been further provided by Kim and Tseng [
7]. Briefly, the amniotic membrane derived from the inner layer of the placenta is composed of an avascular stromal layer and a basement membrane. The amniotic membrane serves as a frame for the migration of corneal epithelium and contains many proteins and anti-inflammatory cytokines that prevent epithelial apoptosis [
8-
12]; furthermore, it does not trigger immunologic rejection [
9,
10]. Conventional amniotic membrane patches (temporary AMT) or grafts (permanent AMT) combined with traditional suture techniques have been proven to be effective in treating various corneal diseases [
13-
15]. However, in contrast to sutureless AMT, these techniques often necessitate the use of an operating room. Sutureless AMT has recently been proposed as a possible substitute for conventional AMT [
16-
20]; this procedure can be easily performed on an outpatient basis and is thus advantageous for elderly patients who have difficulty with complicated processes and preoperative preparations. To the best of our knowledge, only a few studies have evaluated the outcomes of in-office sutureless AMT in PED, and research has been especially sparse with regard to dehydrated amniotic membranes (as compared with cryopreserved amniotic membranes). In the current study, we investigated the potential role of in-office sutureless AMT combined with therapeutic contact lens (T-lens) application for treating PED in various corneal diseases by evaluating the factors affecting treatment success and failure.
Materials and Methods
This study was approved by the institutional review board of Seoul National Bundang Hospital (No. B-2105-682-104) and was performed in accordance with the tenets of the Declaration of Helsinki and its later amendments. Informed consent was exempted for the current study with the approval of the institutional review board of Seoul National Bundang Hospital because of the investigationŌĆÖs minimal risk to participants.
We retrospectively reviewed the medical records of consecutively presenting patients with PED in various corneal diseases who underwent sutureless AMT with T-lens application in an outpatient clinic at Seoul National Bundang Hospital between March 2018 to March 2020. PED diagnoses were confirmed by a corneal specialist (HSJ) using slit lamp microscopy with fluorescein staining. As an inclusion criterion, the definition of PED was as follows: the size of the epithelial defect (ED) either decreased minimally or did not decrease at all despite appropriate conventional treatment for over a week, the margins were heaped up, and the underlying subepithelium or anterior stroma showed opacification. To effectively analyze the factors associated with treatment failure for AMT, we did not exclude any failed cases. Conventional medical treatments prior to AMT for the patients enrolled in our study mainly comprised preservative-free artificial tears and empirical or targeted antimicrobial medications according to antibiotic susceptibility, if causative microbial culture results were confirmed (
Table 1).
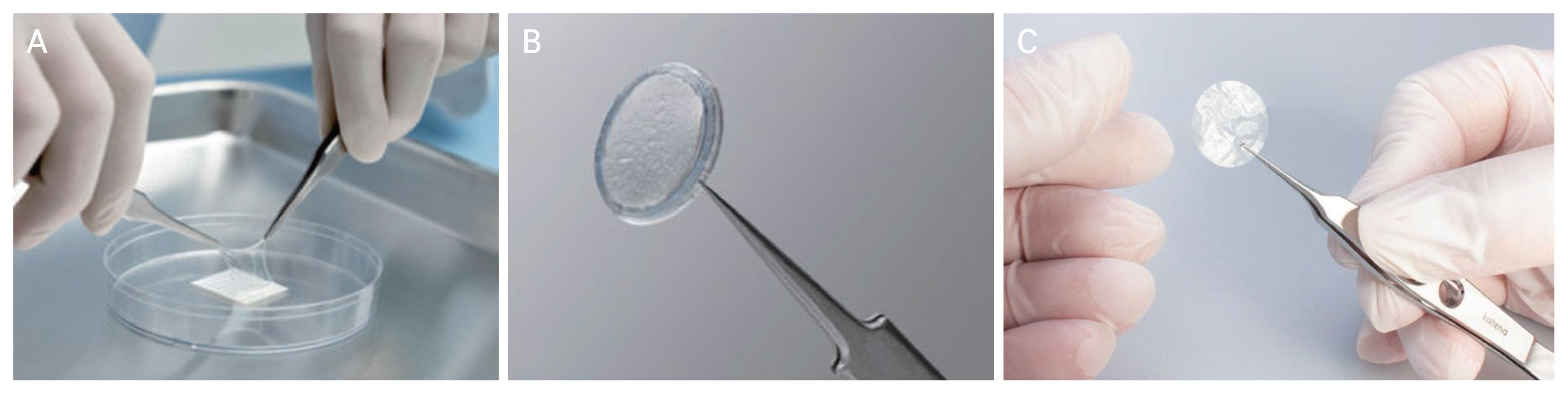
The amniotic membrane used in this study was the AmbioDisk (MiMedx Group, Marietta, GA, USA), which is a dehydrated amniotic membrane graft designed for ophthalmic in-office application. The thickness of the Ambio-Disk is approximately 35 ╬╝m and it has a round shape that is retained even after pinching with mircroforceps. This morphologic feature differs from that of conventional amniotic membranes (which are non-self-retaining and are paper attached) and other cryopreserved sutureless amniotic membranes (i.e., those adhering to a polymethyl methacrylate ring), enabling AmbioDisk to be used more conveniently and effectively in outpatient clinic settings (
Fig. 1A-1C).
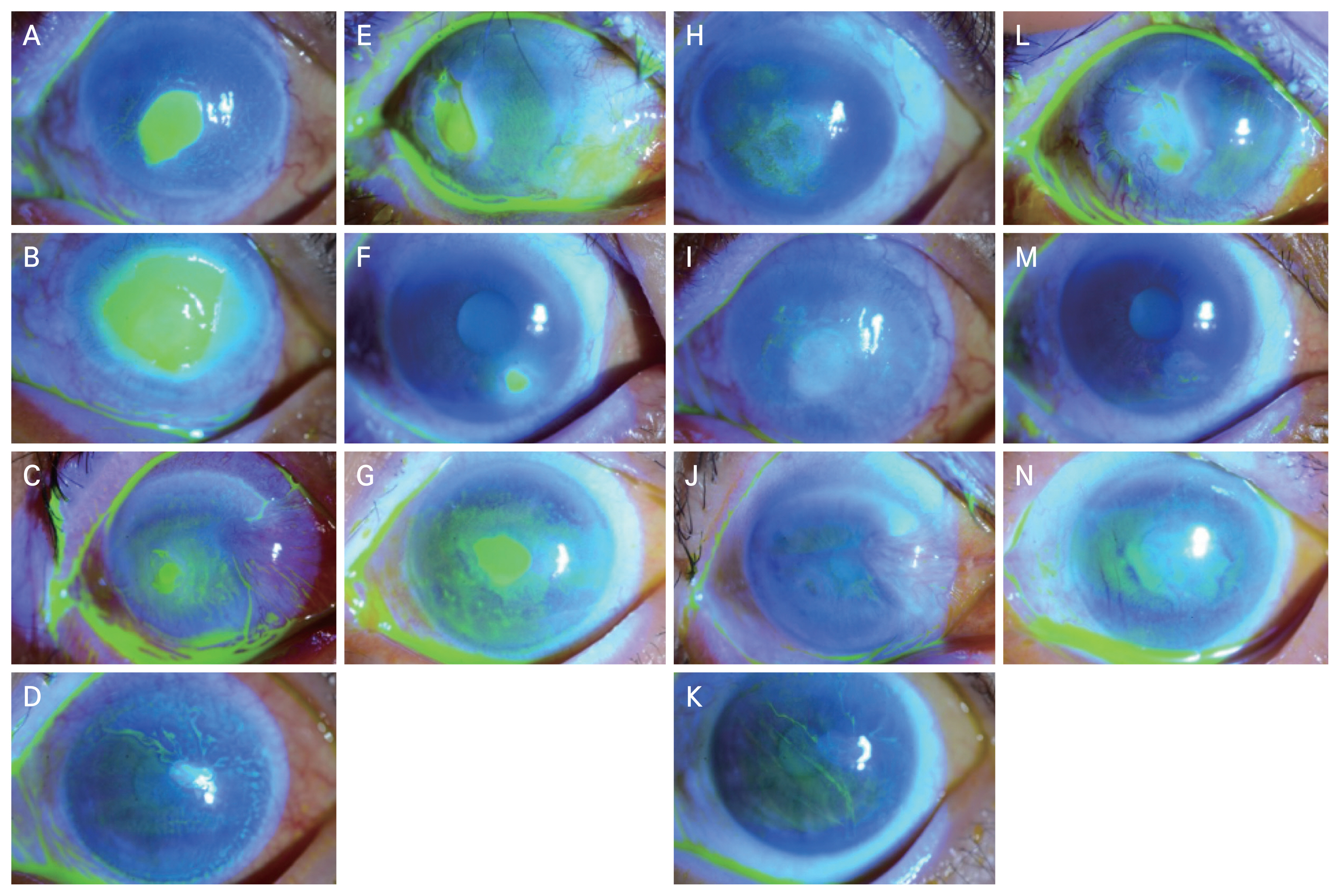
In the current study, after gently applying the Ambio-Disk to the cornea with McPherson forceps, a T-lens (Acuve Oasys; Johnson & Johnson Vision, Jacksonville, FL, USA) was additionally applied over the AmbioDisk in order to stabilize it. Care was taken to dehydrate the ocular surface prior to the removal of the speculum and patients were instructed to apply only one drop of each eye drop solution in order to prevent dislodgment of the amniotic membrane from the ocular surface. Although the T-lens (Acuve Oasys) is generally recommended for up to 2 weeks of continuous use, we maintained the T-lens in order to prevent the amniotic membrane from dislodging until we removed the T-lens after epithelial healing. Representative serial anterior segment photographs taken from the time of AMT until epithelial healing are presented in
Fig. 2A-2J.
After AMT treatment, the use of artificial tears and topical prophylactic antibiotics is recommended three or four times a day in noninfectious cases. If infectious causes could not be ruled out, we maintained the use of pre-AMT medications. Before confirming microbial culture results, frequent application of empirical topical antibiotics (ranging from four times a day to every hour) was recommended. However, if the frequent use of medication was considered harmful for maintaining amniotic membranes, less frequent use of topical treatment was recommended in order to maintain the stability of the amniotic membrane.
The following information was collected from medical records: age, sex, systemic diseases, past ocular history (including PED etiology, corneal ED size, visual acuity before and after AMT, corneal scraping culture results, duration from PED detection to treatment, and duration of healing from PED), and clinical outcomes. The success and failure of each trial of AMT with T-lens application was evaluated according to the following outcome criteria: AMT was regarded as successful when complete epithelial healing was achieved without worsening signs and was regarded as a failed treatment when epithelial healing was inadequate. In these cases, either additional therapy was recommended, or AMT with T-lens application was deemed intolerable due to various causes (and follow-up treatment was hence not recommended), regardless of corneal epithelial status.
Results
The demographics, underlying ocular diseases, clinical features, and outcomes of the patients with PED are summarized in
Table 1. A total of 12 trials in nine eyes (among nine patients) were reviewed in this study. Four male patients and five female patients with a mean age of 71.7 ┬▒ 5.2 years (range, 61-84 years) at the time of AMT with T-lens application were included. The PED etiologies of the enrolled patients included neurotrophic keratopathy (three eyes), infectious keratitis (fungal, three eyes; bacterial, one eye), limbal deficiency related to contact lenses (one eye), and marginal keratitis (one eye). Among the nine eyes, two (cases 1 and 9) underwent more than one trial of AMT with T-lens therapy due to recurrence of PED (case 1) and AMT displacement (case 9).
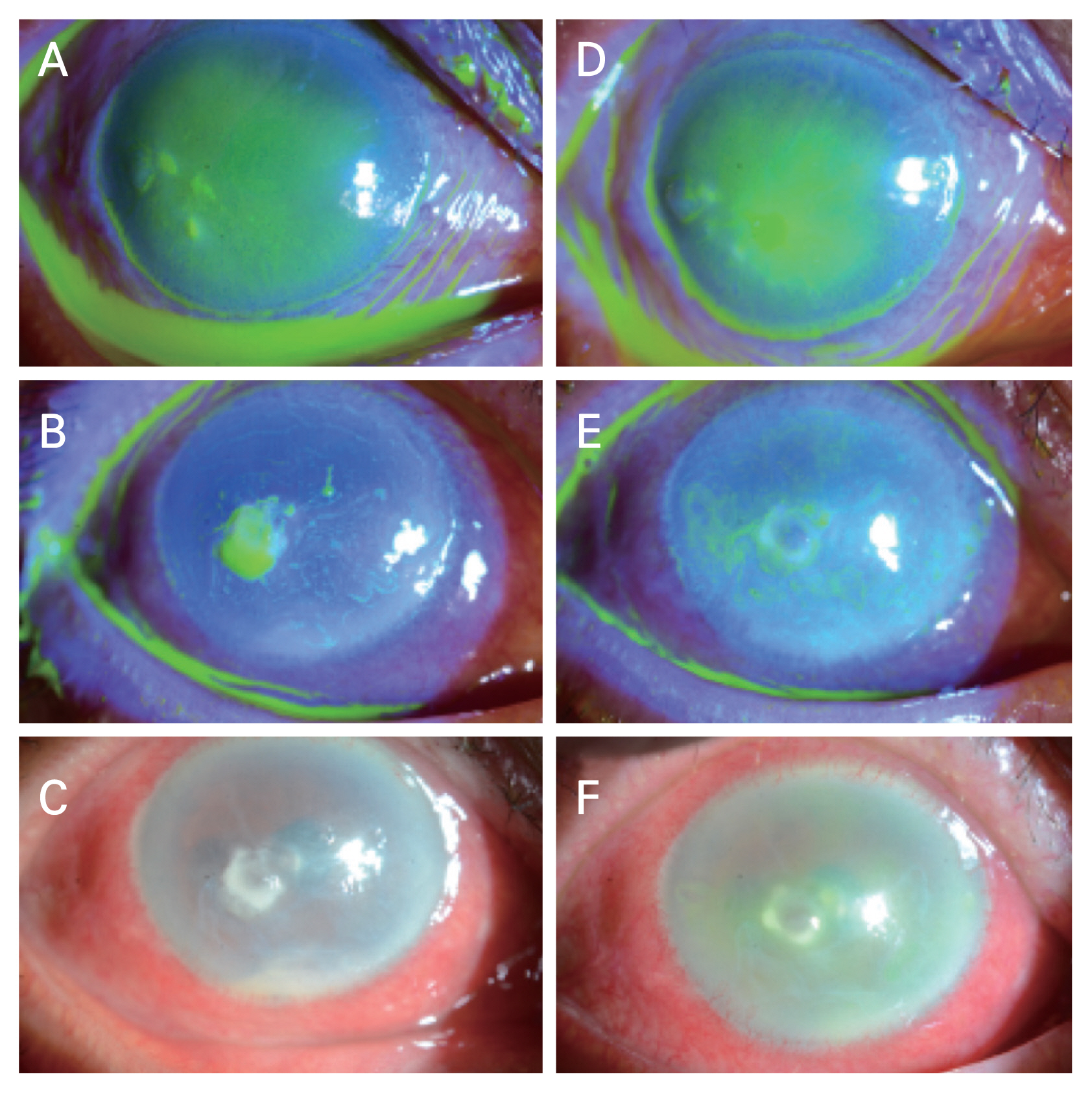
The mean size of corneal ED, which was calculated as the average of the horizontal and vertical diameters measured at the time of AMT with T-lens application, was 3.13 ┬▒ 1.42 mm (range, 0.70-8.25 mm). The mean duration from the detection of ED to AMT with T-lens application was 17.5 ┬▒ 9.7 days (range, 7-37 days). Three trials of AMT with T-lens application were performed in case 9 due to AMT displacement; the follow-up period between AMT application and displacement was 3 days in the first trial and 4 days in the second trial, and both trials occurred over the course of <7 days. Hence, the duration of pre-AMT conventional therapy was calculated as a single trial. Anterior segment photographs before and after sutureless AMT with T-lens application in both successful and failed treatment cases are presented in
Fig. 3A-3N and
Fig. 4A-4F, respectively.
The success rates of AMT with T-lens application within PED were 66.7% (8/12) per trial and 77.8% (7/9) per patient. The mean maintenance duration of AMT with T-lens application was 27.1 ┬▒ 13.0 days (range, 12-51 days) and the mean epithelial healing time following AMT was 30.1 ┬▒ 10.5 days (range, 14-51 days) in successful trials.
In case 7, a patient with neurotrophic keratopathy related to the long-standing use of diclofenac sodium, topical steroids, and glaucoma medications (due to uncontrolled macular edema in uvetic glaucoma) complained of pain after AMT, and AMT with T-lens application was ineffective after 4 days. We hence judged that AMT with T-lens application was ineffective in this case. In case 8, although the corneal epithelium was healed 10 days after AMT treatment, conjunctival injection continued, indicating an uncontrolled infection. This case was thus regarded as a failed treatment according to the pre-specified outcome criteria, and the use of the T-lens was discontinued thereafter. Case 9 presented with exposure keratopathy related to a history of lid reconstruction with reverse Hughes flap surgery due to sebaceous gland carcinoma. Although we recognized that this patientŌĆÖs infectious keratitis was not controlled, we tried AMT with T-lens application with the hope that the PED would improve; however, AMT could not be maintained until the third trial of AMT with temporary tarsorrhaphy.
Discussion
AMT has long been recognized as an effective treatment option for PED. AMT with conventional suture technique has already been demonstrated to be effective in treating various corneal diseases (especially PED), with success rates ranging from 64% to 91% [
21-
23]. However, the conventional suture technique is an invasive and time-consuming procedure, thus necessitating the development of a new, less invasive, and simpler method. With the development and understanding of AMT processing, in-office sutureless AMT has been proposed as a substitute for the conventional suture technique. This treatment modality is divided into cryopreserved and dehydrated AMT.
In 2013, Suri et al. [
19] confirmed success rates of 44% in nonhealing corneal ulcers (nine eyes) and 64% in neurotrophic keratitis (11 eyes) using cryopreserved in-office sutureless AMT (ProKera; Bio-Tissue, Miami, FL, USA). Recently, Mimouni et al. [
20] reported favorable outcomes for in-office sutureless dehydrated AMT (BioDOPTIX; Labtician Ophthalmics, Oakville, ON, Canada) in nine eyes with PED of various etiologies, with an overall success rate of 89% (8/9 eyes). In our study, the success rates of in-office sutureless AMT with T-lens application (per trial) were 75.0% for neurotrophic keratitis and 50% for nonhealing corneal ulcers in infectious keratitis; our results hence varied according to PED etiology and were similar to the favorable outcomes observed in a study of neurotrophic keratopathy conducted by Suri et al. [
19]. A patient with limbal deficiency related to contact lenses and marginal keratitis who had underlying autoimmune hepatitis and thyroid disease also demonstrated successful results at their first AMT trial. The lower overall success rate observed in our study as compared to the study by Mimouni et al. [
20] could be related to differing causes of PED among the included cases; for example, nearly half of the PED patients included in our study presented with infectious keratitis. Although sutureless AMT with T-lens can be applied easily and efficiently in elderly patients with PED (regardless of etiology) on an outpatient basis, we suggest that this treatment may be more effective for treating PED with noninfectious causes, such as neurotrophic keratopathy.
The mean PED healing duration in our study was 30.1 ┬▒ 10.5 days (range, 14-51 days) within successful trials. A previous study by Suri et al. [
19] reported that the mean time to dissolution of the amniotic membrane was 21.3 ┬▒ 29.3 days (range, 2-103 days) in nonhealing corneal ulcers, 11.2 ┬▒ 10.4 days (range, 3-42 days) in the neurotrophic keratopathy group, 15.3 ┬▒ 5.0 days (range, 9-21 days) in the chemical injury group, and 12.9 ┬▒ 10.7 days (range, 7-42 days) among the other participants; these researchers demonstrated that the longest recovery time occurred in PED with infectious causes. Mimouni et al. [
20] reported a mean PED resolution time of 17.8 ┬▒ 9.6 days (range 7-35 days). The longer healing duration observed in our study could be attributed to the higher number of cases with infectious causes as well as the different ED sizes included in the current investigation.
We evaluated characteristic causes of AMT failure in failed trials. The causes of treatment failure in our study were AMT displacement and uncontrolled infection. T-lenses are used indispensably in in-office sutureless AMT in order to stabilize the amniotic membrane, and this could aggravate corneal infection by serving as a bed for microbial infection [
23]. Based on our results, we believe that more careful observation of the signs and symptoms of ocular infection, as well as an additional close follow-up with strict usage of antibiotics or antifungals, is essential to proper treatment. If signs of infection are observed, such as conjunctival injection or aggravation of eye pain, AMT and T-lens application should be discontinued promptly.
Early displacement of the amniotic membrane is a major problem even with the conventional suture method and the use of protective contact lenses [
22]. The displacement rate is an important factor as well, especially in sutureless AMTs with T-lenses, and is expected to be inferior to that observed within the conventional suture method. In our cases, the overall displacement rate was 25% (3 out of 12 trials). Considering other complications related to sutures as well as the accessibility of the operation, this displacement rate seems tolerable. Case 9 had a history of lid reconstruction surgery and PED due to uncontrolled bacterial keratitis; the patient finally succumbed after temporary tarsorrhaphy in the third trial. Since the periocular environment could be an important factor for sutureless AMT stabilization, a careful assessment of the periocular environment as well as appropriate pre-AMT intervention should be performed prior to sutureless AMT with T-lens application if necessary.
Mimouni et al. [
20] reported on patients treated with dehydrated sutureless AMT; specifically, BioDOPTIX for PED demonstrated complete absorption of the dehydrated amniotic membrane within less than 2 weeks in that study. The researchers explained that the shorter absorption time compared to that of ProKera (which has been reported to take up to 125 days to absorb) could be related to the reduced thickness of the dehydrated BioDOPTIX (40 ╬╝m) as compared to the cryopreserved ProKera (100-200 ╬╝m) [
20,
24]. In our study, all cases except two cases with spontaneous displacement of AMT with T-lens application had their AMT with T-lens application removed by a study author (HSJ) after epithelial healing was detected; the amniotic membrane was maintained until the T-lens was removed in all these cases. We considered that the absorption time may be longer than the duration of T-lens treatment (27.1 ┬▒ 13.0 days; range, 12-51 days) within our case series. We present representative anterior segment photographs of two cases before, during, and after AMT in
Fig. 2. As the amniotic membrane (AmbioDisk) is thin and translucent, the configuration of the membrane under the T-lens cannot be fully observed accurately. We expect similar absorption time between AmbioDisk and BioDOPTIX as the thickness of the amniotic membrane is approximately 40 ╬╝m for both treatment modalities. Features such as thickness and absorption time may be important considerations for choosing amniotic membrane types according to the ocular environment. Further investigation regarding absorption time for the AmbioDisk within a larger number of cases is necessary in future research.
This study has some limitations in addition to its substantial strengths. First, the number of cases enrolled in this study was small, our study enrolled PED cases with heterogenous causes, and there was no control group. These limitations are inherent to all retrospective studies. Since PED is a rare condition, it is difficult to plan well-controlled studies evaluating PED treatments. However, our case series, along with other prior studies on sutureless AMT will be informative to clinicians encountering patients with refractory PED. Additional case series will aid in the establishment of indications for the treatment of PED, including based on size, thus allowing this expensive treatment to be performed more successfully. Second, in-office sutureless dehydrated AMT inevitably requires a T-lens in order to stabilize the amniotic membrane, which makes it difficult to distinguish whether T-lenses or AMTs with T-lenses are affecting epithelial healing. Epithelial healing may be a protective effect of T-lenses; however, since we included PED cases that were refractory to conventional treatment for over a week, our findings suggest that the amniotic membrane may have additive anti-inflammatory and epithelial healing promotive effects. To make study results more accurate and distinguishable, a further comparative case-control study with a T-lens control group (versus AMT with a T-lens group) will be necessary in future research efforts. Third, this study did not include accurate information regarding amniotic membrane melting time. Further, we did not exchange T-lenses until the amniotic membrane melted for the purpose of stabilizing the amniotic membrane. Long-standing use of a single T-lens could increase the chance of infection by providing a microbial bed and could thus be harmful for patients if not applied with adequate consideration of the amniotic membrane melting time. Studies evaluating more accurate timing with regard to amniotic membrane melting time may be helpful for reducing infection risk by providing adequate follow-up time as well as minimizing T-lens continuation.
We believe that in-office sutureless AMT is particularly helpful for elderly patients who often have difficulty with the preoperative general examination to enter the operating room while aiding in economic and efficiency optimization. Moreover, neurotrophic keratopathy (which is a main cause of PED) is prevalent in elderly patients and is a good indication for sutureless AMT. However, we conclude that this treatment modality cannot replace conventional amniotic membrane patches and grafts as it may be difficult to maintain due to the influence of the surrounding ocular environment (eyelids or conjunctiva). There may also be a possibility that the sutureless membrane will move with blinking. According to our experience, we recommend that in-office sutureless AMT be used in cases where the defect size is less than half of the cornea, there is no eyelid defect, and infection is under control.
In conclusion, the results of our case series demonstrate that in-office sutureless AMT combined with T-lens application may be a simple and effective treatment in PED patients refractory to medications, particularly among elderly patients. An appropriate periocular environment (including consideration of the eyelids) and infection control are important to achieving a successful in-office sutureless AMT with T-lens application in refractory cases of PED.






 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print




