 |
 |
| Korean J Ophthalmol > Volume 33(2); 2019 > Article |
Abstract
Purpose
To evaluate laser in situ keratomileusis (LASIK) flap thickness predictability and morphology by femtosecond (FS) laser and microkeratome (MK) using anterior segment optical coherence tomography.
Methods
Fifty-two candidates for the LASIK procedure were stratified into two groups: FS laser-assisted (Allegretto FS-200) and MK flap creation (Moria 2). Flap thickness was determined at five points. The side-cut angle was measured in three directions at the margin interface. LASIK flap assessment was performed one month postoperatively by Spectralis anterior segment optical coherence tomography.
Results
Fifty-two patients (93 eyes) were recruited; 49 eyes were stratified to the FS group and 44 eyes to the MK group. The FS group had relatively even flap configurations, and the MK group had meniscus-shaped flaps. Mean differences between planned and actual flap thickness were 12.93 ┬▒ 8.89 and 19.91 ┬▒ 5.77 ┬Ąm in the FS and MK groups, respectively. In thin flaps (100 to 110 ┬Ąm), there was a significant disparity between the two groups (7.80 ┬▒ 4.71 and 19.44 ┬▒ 4.46 ┬Ąm in the FS and MK groups, respectively). However, in thicker flaps (130 ┬Ąm), comparable flap thickness disparity was achieved (18.54 ┬▒ 9.52 and 20.83 ┬▒ 5.99 ┬Ąm in the FS and MK groups, respectively). Mean side-cut angle was 74.29 ┬▒ 5.79 degrees and 32.34 ┬▒ 4.94 degrees in the FS and MK groups, respectively.
Corneal refractive surgery has shown remarkable progress during the last decade, with fast-growing updates in operative techniques, devices, and instrumentation. However, laser in situ keratomileusis (LASIK) remains the most common corneal refractive procedure [1]. Flap creation is the most critical step during LASIK eye surgery, so the consistency and predictability of the corneal flap thickness are crucial for a successful LASIK outcome. Thus, improving the predictability of LASIK flap thickness and morphology is worthy of attention [2].
In mechanical microkeratome (MK)-assisted flap creation, an oscillating blade is used to create corneal flaps. Despite the advances in MK designs, flap-related complications, such as free caps, button holes, incomplete cuts, and torn flaps, remain a challenge for most refractive surgeons [3,4]. In femtosecond (FS)-assisted flap creation, a cleavage line is created through the cornea at a predetermined depth by photo-ionization of optically transparent tissues with a resultant acoustic shock wave and gas bubble formation, disrupting treated tissues [5].
Many FS laser systems are available now, including IntraLase (Abbott Medical Optics, Santa Ana, CA, USA), VisuMax (Carl Zeiss Meditec AG, Dublin, CA, USA), Femto LDV (Zeimer Group, Port, Switzerland), and FS 200 WaveLight (Alcon Laboratories, Fort Worth, TX, USA) [6]. Different devices are available for measuring postoperative flap thickness depending on subtraction from the preoperative planned residual stromal bed; however, these measurements are rough and mostly inaccurate [7,8]. Using real-time images, anterior segment optical coherence tomography (AS-OCT) is now the most widely-used corneal imaging system not only to assess post-LASIK flap thickness, but also to evaluate flap morphology and determine the side cut angle at the flap margin interface [9,10]. The objective of our study was to assess LASIK flap thickness predictability created by a FS laser versus a mechanical MK, in addition to flap morphology and side cut angle using AS-OCT.
A prospective, comparative, open-label study was conducted from January 2016 to April 2016 after approval of the ethical committee of the Faculty of Medicine, Assiut University, Egypt (0032016) and under the tenets of Helsinki declaration. A written informed consent was signed by all patients after a thorough explanation of the procedure and its possible complications.
The inclusion criteria were myopic LASIK correction up to ŌłÆ12 diopters with or without myopic astigmatism up to ŌłÆ6 diopters, a corneal thickness at the thinnest location Ōēź500 ┬Ąm, and a residual stromal bed Ōēź280 ┬Ąm. Exclusion criteria were hyperopic refraction, mixed astigmatism, systemic disease that contraindicates LASIK, and intraoperative or postoperative complications.
All candidates underwent detailed history-taking and complete ophthalmic examination, including uncorrected visual acuity (UCVA) and best-corrected visual acuity by Landolt's C-chart, slitlamp biomicroscopy (Haag-Streit, Mason, OH, USA), intraocular pressure measurements (Goldmann Applanation tonometer mounted on a slitlamp), Schirmer I test, and Pentacam evaluation (Oculus Pentacam, Oculus GmbH, Wetzlar, Germany).
Candidates were stratified into two groups according to flap creation technique: FS or MK. In the FS group, patients underwent FS-assisted flap creation using Allegretto WaveLight FS-200 FS laser (Alcon Laboratories). The device used a 200-kHz repetition rate, 1,030-nm wavelength, and 5-┬Ąm spot size. The settings of the flap creation procedure were set so that the hinge was superior with a fixed flap diameter of 9 mm and a side cut angle of 70 degrees in all patients. The planned flap thickness was subdivided into 100 to 110 ┬Ąm and 130 ┬Ąm groups according to the patient's corneal and refractive profile and according to surgeon's preference.
In the MK group, patients underwent mechanical flap creation using a Moria 2 Microkeratome (Moria SA, Antony, France), where flaps with superior hinges were created with variable diameters according to keratometric readings. Planned flap thickness was also subdivided into 100 to 110 ┬Ąm and 130 ┬Ąm groups according to the surgeon's preference after choice of a suitable suction ring.
Myopic laser ablation was performed using EX-500 excimer laser (Alcon Laboratories) with a planned full correction and post-operative emmetropia. All cases were done by the same experienced surgeon (MA) in both groups. Post-LASIK follow-up was scheduled 1-day, 1-week, 2-week, and 1-month post-LASIK. A combination of Dexamethasone-Tobramycin eye drops (Tobradex, Alcon Laboratories) four times daily for 1 week and topical lubricant eye drops (Systane Ultra, Alcon Laboratories) four times daily for 3 months were prescribed.
All patients underwent anterior segment optical coherence tomography (AS-OCT) evaluation of flap morphology, thickness, and side-cut by Spectralis spectral domain optical coherence tomography (Heidelberg Engineering, Heidelberg, Germany) 1-month post-LASIK. The Spectralis AS-OCT had an acquisition speed of 40,000 A-scans per second, with an axial resolution of 3.9 to 7 ┬Ąm and a transverse resolution of 14 ┬Ąm. Flap assessment by AS-OCT was carried out by a single experienced ophthalmologist (ZE). Flap thickness was measured at five points along the horizontal meridian passing through the corneal center. The corneal center was determined by the presence of high reflective artifacts while scanning for corneal apex. The flap was evaluated with a horizontal line scan measuring 20 degrees and averaged to 10 frames only to avoid overexposure and loss of LASIK interface details. The LASIK flap thickness was measured at five points: center, 1 mm nasal and temporal, and 2.5 mm nasal and temporal. Flap thickness was defined as the distance between the tear film-epithelial interface and the LASIK flap interface and perpendicular to tear film-epithelial interface. It was measured three times, and the average was calculated. The adjustments were done manually, and readings were calculated accordingly based on the measuring tool of the device. For side-cut angles, a 15-degree line scan averaged to 10 frames was aligned perpendicularly across nasal, temporal, and inferior edges of the flap to acquire scans across the three flap edges. Images were then t ransferred to ImageJ software ver. 1.8.0 (National Institutes of Health, Bethesda, MD, USA) [11]. The angle measuring tool in the software was utilized to measure the side-cut angles three times, and the mean reading was recorded by a single experienced ophthalmologist (MN).
The statistical analysis was done by IBM SPSS Statistics ver. 20.0 (IBM Corp., Armonk, NY, USA). Descriptive statistics were evaluated to compare patient characteristics between groups. Mann-Whitney test was used to compare means among groups. A p-value less than 0.05 was considered significant.
This study included 93 eyes of 52 patients. FS-assisted LASIK surgery was done in 49 eyes of 30 patients (14 males and 16 females), while 44 eyes of 22 patients (6 males and 16 females) underwent LASIK surgery with MK-assisted flap creation. Table 1 demonstrates the baseline patient characteristics.
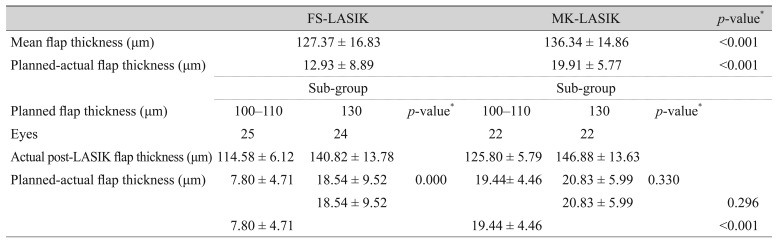
Mean f lap thickness was 127.37 ┬▒ 16.83 ┬Ąm in FS-LASIK patients and 136.34 ┬▒ 14.86 ┬Ąm in MK-LASIK patients. There was a significant difference between planned and actual flap thickness between the two groups regarding the overall mean f lap thickness measurement (p < 0.001) (Table 2).
Furthermore, planned flap thickness was categorized in each group into two sub-groups: 100 to 110 ┬Ąm and 130 ┬Ąm planned flap thickness. In the FS-LASIK group, planned flap thickness of 100 to 110 ┬Ąm was significantly more predictable than 130 ┬Ąm (p < 0.001), with planned-actual flap thickness of 7.80 ┬▒ 4.71 and 18.54 ┬▒ 9.52 ┬Ąm, respectively. Meanwhile, in MK-LASIK patients, there was no significant difference between 100 to 110 ┬Ąm and 130 ┬Ąm planned flap thickness (p = 0.330), with planned-actual flap thickness of 19.44 ┬▒ 4.46 ┬Ąm and 20.83 ┬▒ 5.99 ┬Ąm, respectively.
FS-LASIK yielded a significantly more predictable flap thickness when comparing planned 100 to 110 ┬Ąm flap thickness in both treatment groups (planned-actual flap thickness, 7.80 ┬▒ 4.71 and 19.44 ┬▒ 4.46 ┬Ąm in FS-LASIK and MK-LASIK groups, respectively, p < 0.001). On the other hand, the two treatment groups yielded a comparable flap thickness when planned flap was adjusted for 130 ┬Ąm with insignificant difference (18.54 ┬▒ 9.52 and 20.83 ┬▒ 5.99 ┬Ąm in FS-LASIK and MK-LASIK groups, respectively, p = 0.296).
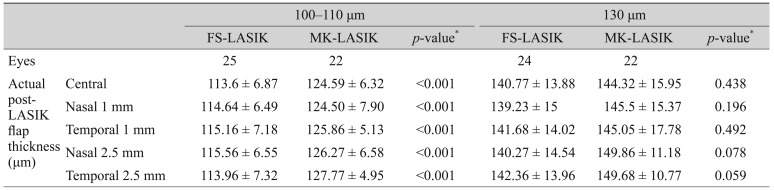
Mean flap thicknesses at five different points (center, 2.5 mm nasally, 1 mm nasally, centrally, 1 mm temporally, and 2.5 mm temporally) are demonstrated i n Table 3. T he point-to-point difference with regard to treatment groups and planned flap thickness agreed with mean flap thickness difference between the two groups.
In the FS-LASIK groups, there was a relatively even flap configuration where mean flap thickness was nearly equal when measured at five different points across the horizontal meridian, while in the MK-LASIK group, the LASIK flap had a meniscus configuration, being thinnest at the center and gradually increasing in thickness toward the periphery (Fig. 1A, 1B).
This meniscus configuration in MK-LASIK was best demonstrated when planned flap thickness was 130 ┬Ąm, while a less prominent meniscus configuration was found 100 to 110 ┬Ąm planned flap thickness.
LASIK flaps in the FS group had a mean side-cut angle of 74.29 ┬▒ 5.79 degrees (range, 59 to 84 degrees; 95% confidence interval [CI], 72.62 to 75.95 degree,) while the MK group had a mean side-cut angle of 32.34 ┬▒ 4.94 degrees (range, 23 to 41 degrees; 95% CI, 30.84 to 33.85 degree). As FS-LASIK flap side-cut angle was pre-operatively adjusted to 70 degrees, mean difference between planned and actual post-LASIK flap side-cut was 6.12 ┬▒ 3.73 degrees (range, 0 to 14 degrees; 95% CI, 5.05 to 7.2 degree).
In AS-OCT evaluation of side-cut angle, FS-LASIK side-cut edges were well-demarcated and regularly outlined, while MK-LASIK side-cut edges showed a ragged outline in many cases (Fig. 2A-2C).
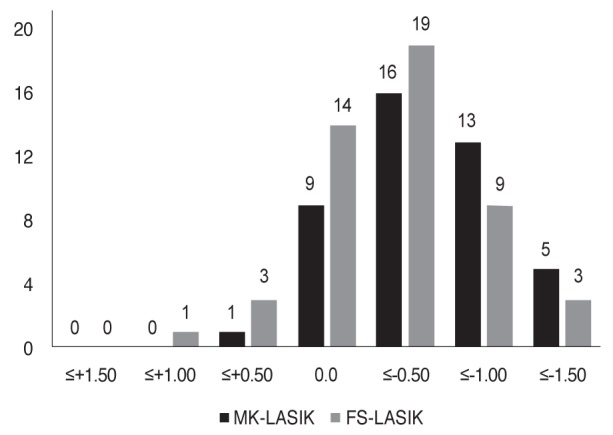
Mean post-LASIK manifest refraction spherical equivalent (MRSE) were ŌłÆ0.43 ┬▒ 0.62 and ŌłÆ0.49 ┬▒ 0.46 diopters in FS-LASIK and MK-LASIK groups, respectively, with significant difference from pre-LASIK MRSE (p < 0.001). There was a tendency for MK-LASIK to induce slightly more myopic post-LASIK refraction than FS-LASIK after one month; however, there was no significant impact on post-LASIK UCVA (Fig. 3).
In this prospective study, we investigated the predictability of LASIK flap thickness and its configuration by two different methods, FS- and MK-assisted flap creation. We analyzed LASIK f lap characteristics and measurements by AS-OCT. Many clinical studies have highlighted the utility of AS-OCT in the evaluation of LASIK flaps created by either MK or FS laser [10,12].
We evaluated the LASIK flap at five different points along the horizontal meridian of the cornea rather than a single central corneal point, as used in other studies [13], to provide a comprehensive overview of the LASIK flap and its configuration both qualitatively and quantitatively. In addition, flap configuration and peripheral architecture should not be overlooked. Planned LASIK flap thickness was stratified into two subgroups: 100 to 110 ┬Ąm and 130 ┬Ąm. Our study highlighted the predictability of flap thickness by MK and FS laser in both thin flaps (110 to 110 ┬Ąm) and thicker flaps (130 ┬Ąm) to spotlight the advantage of FS-assisted LASIK. Other studies have overlooked such stratification, and different planned flap thicknesses were pooled together, neglecting the possible difference in flap thickness. Thereafter, a separate comparison was carried out between planned and actual post-LASIK flap thickness in both groups. Such a detailed comparison clarifies the different performances of FS- and ML-assisted flap creation in different planned flap thickness. Regarding mean f lap thickness in pooled data, there was a significant planned-actual flap thickness difference between FS- and MK-assisted groups (12.93 ┬▒ 8.89 and 19.91 ┬▒ 5.77 ┬Ąm, respectively, p < 0.001), with less difference in the FS-assisted group. However, after stratifying both groups into 100 to 110 ┬Ąm and 130 ┬Ąm subgroups, different results were produced. In the 100 to 110 ┬Ąm subgroup, there was a significant planned-actual flap thickness difference (7.80 ┬▒ 4.71 and 19.44 ┬▒ 4.46 ┬Ąm, respectively, p < 0.001). The 100 to 110 ┬Ąm actual FS-assisted flap was less than 10 ┬Ąm than planned, reflecting high reproducibility of FS in this flap thickness category. On the contrary, there was no significant planned-actual flap thickness difference in the 130 ┬Ąm subgroup between FS- and MK-assisted flap creation (18.54 ┬▒ 9.52 and 20.83 ┬▒ 5.99 ┬Ąm, respectively, p = 0.296). Therefore, if a surgeon plans a 130 ┬Ąm LASIK flap, there is no preference of FS- over MK-assisted flap regarding flap thickness reproducibility. Moreover, point-to-point comparison revealed a significant difference in all points between FS- and MK-assisted flap creation in the 100 to 110 ┬Ąm subgroup and a non-significant difference in all points in the 130 ┬Ąm subgroup (Table 3).
Flap configuration varied between the two groups. A meniscus flap configuration was obtained in the MK group and was more pronounced at 130 ┬Ąm flap thickness. The FS group yielded a more uniform flap regardless of flap thickness (Fig. 1). This could be explained by the course of the MK through the cornea during flap creation, while in the FS group, flattening produced by a suction cup over the cornea results in uniform FS laser application and flap configuration. Despite the difference in flap configuration, center-periphery difference was not significant in any subgroup analysis, reflecting the reproducibility of both techniques in flap creation. Though flap configuration was a key predictor of flap stability, side-cut angles remain one of the most important factors in flap stability. One of the advantages of FS-assisted flap creation is the ability to adjust the side-cut angle to ensure greater flap stability. In the FS group, planned-actual side-cut angle difference (70-degree planned side-cut angle) was 6.12 ┬▒ 3.73 degrees, highlighting the accuracy of FS-assisted flap creation. Furthermore, the side-cut architecture varied among the two groups. AS-OCT revealed a shaggy side-cut of MK-assisted flap with an acute angle (32.34 ┬▒ 4.94 degrees), while FS-assisted flap showed a uniform angled side cut (74.29 ┬▒ 5.79 degrees), providing a more stable flap. Despite the qualitative and quantitative differences between these two techniques of flap creation, refractive outcomes (as regarding post-LASIK MRSE and UCVA) were similar between FS- and MK-assisted LASIK.
Zhang et al. [14] documented that the difference between planned and actual flap thickness was 5.61 and 31.52 ┬Ąm in MK (Hansatome, Bausch & Lomb, Rochester, NY, USA) and FS (Femto LDV, Ziemer Group) groups, respectively. In another study, the difference between the achieved and the planned flap thickness was 6.17 and 23.60 ┬Ąm in MK (Moria Keratome; Moria SA, Antony, France) and FS (WaveLight FS200, Alcon Laboratories) groups, respectively [15]. Despite the difference in flap thickness predictability, all mentioned studies showed no difference in final refractive outcome between FS and MK groups. However, all previously mentioned studies compared flap thickness reproducibility without stratification of planned flap thickness, which could mislead data interpretation and undermine the comparable accuracy of both techniques in thicker flaps, as documented in our study. On the other hand, Zhou et al. [16] found that deviations greater than 20 ┬Ąm in actual flap thickness (planned flap thickness was 110 ┬Ąm for all patients) were observed more frequently in the MK group than the FS group (42.4% and 0.73% of eyes, respectively) in thin LASIK flaps. Such findings could spotlight the higher accuracy of FS over MK, as presented in our study. Different FS laser machines could provide different flap thickness predictability [10]. The Femto LDV device offered higher predictability than the IntraLase FS and Visumax FS. Meanwhile, in a study conducted by Liu and co-authors, comparable flap thickness reproducibility was achieved by both Intralase FS60 and Wavelight FS200 devices (Alcon Laboratories) [17].
Many studies have highlighted the difference in flap morphology between FS- and MK-assisted LASIK. The architecture of FS-assisted flaps is uniform even with different FS machines [18,19], while MK-assisted LASIK flaps yield a meniscus-shaped architecture, as discussed in other studies [14,15]. One drawback in some FS devices is the limited choices in selection of planned flap thickness [10]. Surgeons should consider these limitations during patient counseling and surgical planning to deliver the safest and most accurate surgical plan. In contrast, MK devices provide a wide selection of planned flap thickness to fit every patient's profile.
Though some FS devices do not have the capability to adjust the side-cut angles, other devices provide such an advantage. The side-cut angle in the IntraLase device was 74.50 degrees when planned to be 70 degrees, reflecting the accuracy of the technique compared to our study (74.29 degrees for planned side-cut a ngle of 70 d egrees) [10]. However, our study did not evaluate f lap stability and flap-related complications, which are yet to be investigated in a large-scale study.
In summary, FS-assisted flap creation yielded a more predictable LASIK flap when a thinner planned flap (100 to 110 ┬Ąm) was intended. However, FS and MK techniques performed equivalently with thicker LASIK f laps (130 ┬Ąm). Nevertheless, flap configuration was an important difference between these two techniques, in addition to side-cut angles, which may have a significant impact on flap stability.
Notes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
REFERENCES
1. Sugar A, Rapuano CJ, Culbertson WW, et al. Laser in situ keratomileusis for myopia and astigmatism: safety and efficacy: a report by the American Academy of Ophthalmology. Ophthalmology 2002;109:175-187.


2. Pietila J, Makinen P, Suominen S, et al. Corneal flap measurements in laser in situ keratomileusis using the Moria M2 automated microkeratome. J Refract Surg 2005;21:377-385.


4. Ambrosio R Jr, Wilson SE. Complications of laser in situ keratomileusis: etiology, prevention, and treatment. J Refract Surg 2001;17:350-379.


5. Kurtz RM, Liu X, Elner VM, et al. Photodisruption in the human cornea as a function of laser pulse width. J Refract Surg 1997;13:653-658.


6. Salomao MQ, Wilson SE. Femtosecond laser in laser in situ keratomileusis. J Cataract Refract Surg 2010;36:1024-1032.



7. Muallem MS, Yoo SH, Romano AC, et al. Flap and stromal bed thickness in laser in situ keratomileusis enhancement. J Cataract Refract Surg 2004;30:2295-2302.


8. Cheng HC, Chen YT, Yeh SI, Yau CW. Errors of residual stromal thickness estimation in LASIK. Ophthalmic Surg Lasers Imaging 2008;39:107-113.


9. Grewal DS, Brar GS, Grewal SP. Assessment of central corneal thickness in normal, keratoconus, and post-laser in situ keratomileusis eyes using Scheimpflug imaging, spectral domain optical coherence tomography, and ultrasound pachymetry. J Cataract Refract Surg 2010;36:954-964.


10. Ahn H, Kim JK, Kim CK, et al. Comparison of laser in situ keratomileusis flaps created by 3 femtosecond lasers and a microkeratome. J Cataract Refract Surg 2011;37:349-357.


11. Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods 2012;9:671-675.



12. Prospero Ponce CM, Rocha KM, Smith SD, Krueger RR. Central and peripheral corneal thickness measured with optical coherence tomography, Scheimpflug imaging, and ultrasound pachymetry in normal, keratoconus-suspect, and post-laser in situ keratomileusis eyes. J Cataract Refract Surg 2009;35:1055-1062.


13. Pajic B, Vastardis I, Pajic-Eggspuehler B, et al. Femtosecond laser versus mechanical microkeratome-assisted flap creation for LASIK: a prospective, randomized, paired-eye study. Clin Ophthalmol 2014;8:1883-1889.


14. Zhang XX, Zhong XW, Wu JS, et al. Corneal flap morphological analysis using anterior segment optical coherence tomography in laser in situ keratomileusis with femtosecond lasers versus mechanical microkeratome. Int J Ophthalmol 2012;5:69-73.


15. Zhang Y, Chen YG, Xia YJ. Comparison of corneal flap morphology using AS-OCT in LASIK with the WaveLight FS200 femtosecond laser versus a mechanical microkeratome. J Refract Surg 2013;29:320-324.


16. Zhou Y, Zhang J, Tian L, Zhai C. Comparison of the Ziemer FEMTO LDV femtosecond laser and Moria M2 mechanical microkeratome. J Refract Surg 2012;28:189-194.


17. Liu Q, Zhou YH, Zhang J, et al. Comparison of corneal flaps created by Wavelight FS200 and Intralase FS60 femtosecond lasers. Int J Ophthalmol 2016;9:1006-1010.



Fig.┬Ā1
Laser in situ keratomileusis (LASIK) flap side-cut angle and edge outline in (A) microkeratome-assisted LASIK and (B) femtosecond laser-assisted LASIK groups by anterior segment optical coherence tomography.
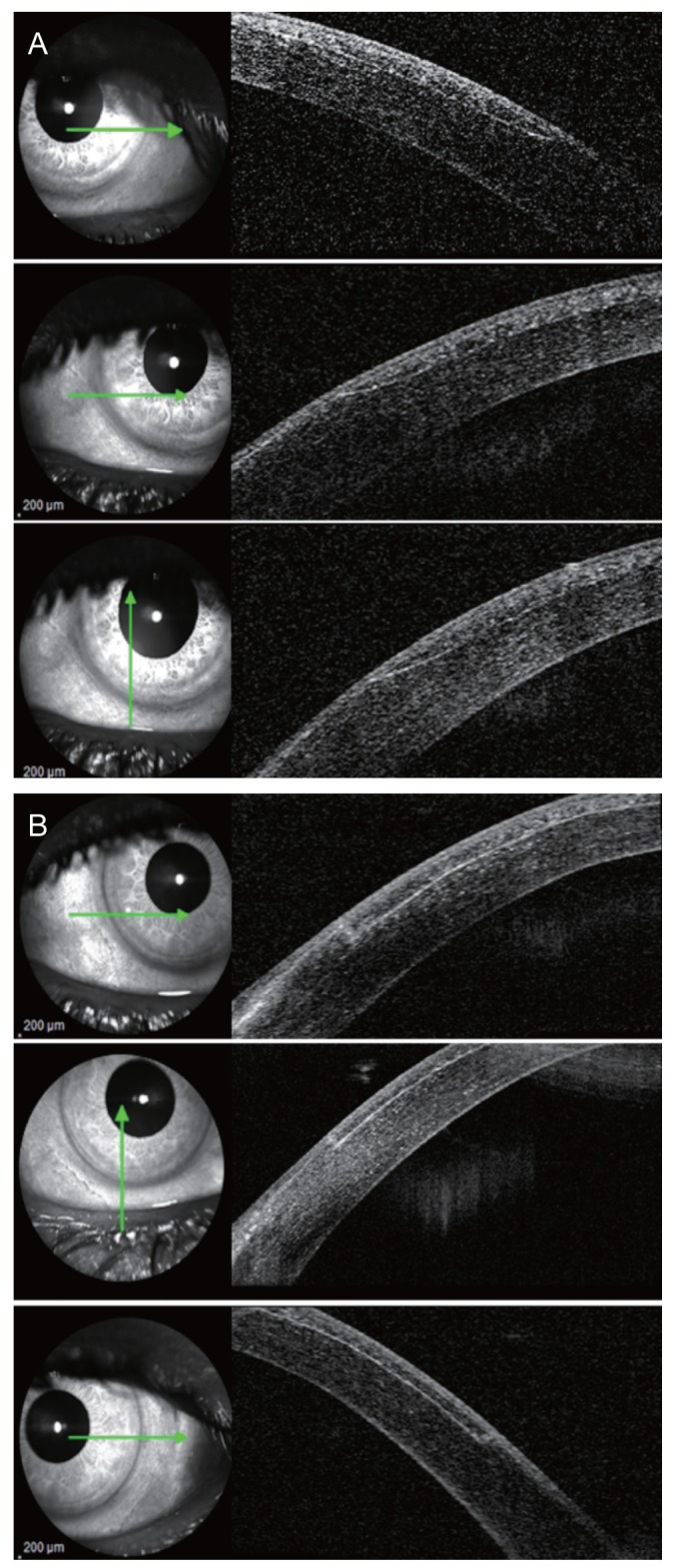
Fig.┬Ā2
Average flap thickness in femtosecond laser-assisted laser in situ keratomileusis (LASIK) and microkeratome-assisted LASIK groups as measured in five different locations (centrally, 1 mm nasally and temporally, 2.5 mm nasally and temporally across horizontal meridian). The difference between two groups is more evident in planned flap thickness of 100 to 110 ┬Ąm. (A) Planned flap thickness (pooled date), (B) planned LASIK flap (130 ┬Ąm), and (C) planned LASIK flap (100 to 110 ┬Ąm). N = nasal; T = temporal.
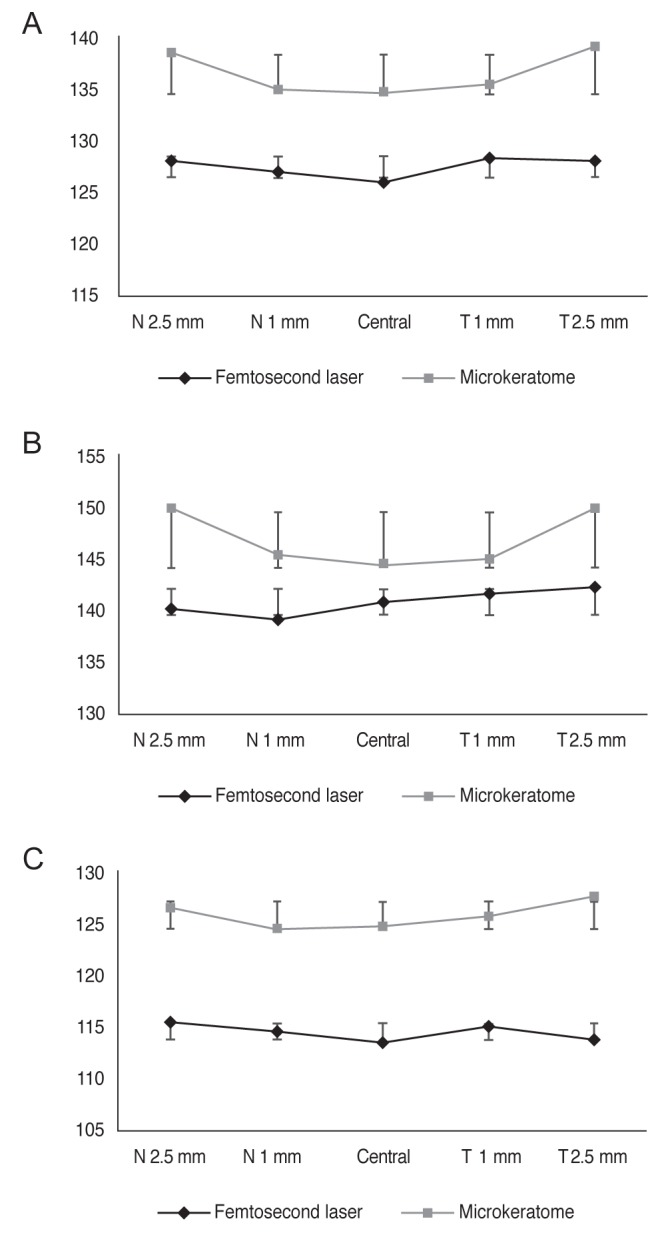
Fig.┬Ā3
Frequency of distribution of postoperative (microkeratome-assisted laser in situ keratomileusis [MK-LASIK] and femtosecond laser-assisted laser in situ keratomileusis [FS-LASIK]) manifest spherical equivalent.
Table┬Ā1
Patient characteristics in both femtosecond laser-assisted LASIK and microkeratome-assisted LASIK

Values presented as mean ┬▒ standard deviation.
LASIK = laser in situ keratomileusis; FS-LASIK = femtosecond laser-assisted LASIK; MK-LASIK = microkeratome-assisted LASIK; MRSE = manifest refraction spherical equivalent; BCVA = best-corrected visual acuity; UCVA = uncorrected visual acuity; CCT = central corneal thickness; K value = keratometric readings.
*Mann-Whitney test.
Table┬Ā2
Planned and actual flap thickness in both femtosecond laser-assisted LASIK and microkeratome-assisted LASIK
- TOOLS
-
METRICS

-
- 6 Crossref
- 0 Scopus
- 2,350 View
- 30 Download
- Related articles




 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



