 |
 |
| Korean J Ophthalmol > Volume 32(6); 2018 > Article |
Abstract
Purpose
Methods
Results
Notes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
REFERENCES
Fig. 1
Measurement of lamina cribrosa depth (LCD) by spectral-domain optical coherence tomography. (A) En-face images of the optic nerve head were obtained from spectral-domain optical coherence tomography enhanced depth imaging scanning. Among several B-scans from optic disc scanning, a horizontal section image passing through major vessel trunks was selected to measure the LCD. This image-selecting procedure was repeated in the same eye at each follow up visit. (B) A line connecting the Bruch's membrane opening (red dots) on each side was regarded as the Bruch's membrane opening plane. The anterior surface of the lamina cribrosa was set as the horizontal margin, where the highly reflective region beneath the optic disc cup started. The distance of the three perpendicular lines (composed of the maximally depressed point, yellow arrow) from the reference line, and 100 and 200 µm apart from the maximally depressed point to temporal direction, were measured. A manual caliper tool and regions of interest manager of ImageJ software was used for each measurement and the LCD was determined by the average value measured from the three points.
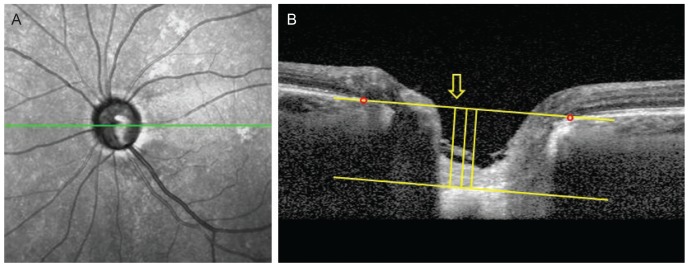
Fig. 2
Subgroup analysis of lamina cribrosa displacement. The method of measuring lamina cribrosa depth (LCD) described in Fig. 1 was repeated at each follow-up visit. The values were determined at each follow-up visit and the difference from the first to the last measurement was considered LCD change. A significant LCD change was defined as a value greater, within and less than the reproducibility coefficient (23.08 µm). (A) Posteriorly displaced lamina cribrosa (LC), (B) no significant LC displacement, and (C) anteriorly displaced LC.
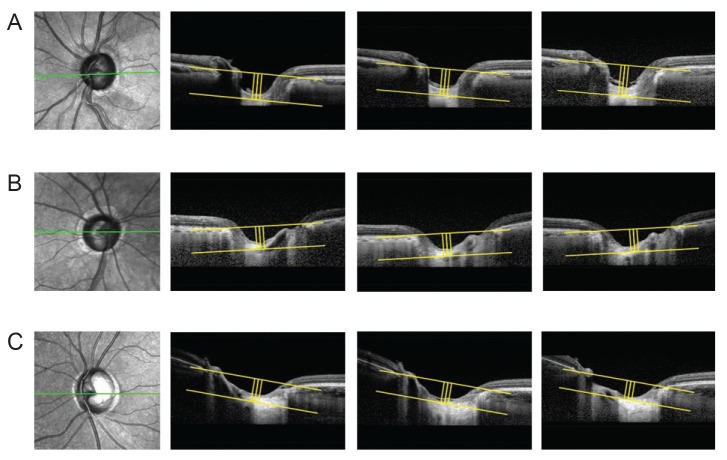
Fig. 3
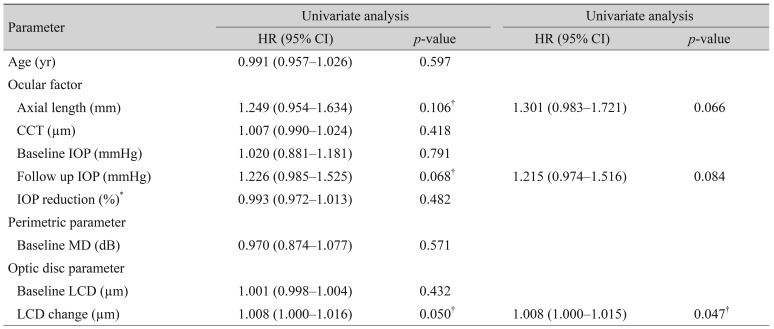
Kaplan-Meier estimates of visual field progression during the follow up period. X-axis, follow-up period (year); Y-axis, cumulative probability of the visual field progression. *p< 0.05 considered as statistically significant. LCD = lamina cribrosa depth; ILCD = increased LCD; NLCD = no LCD change; DLCD = decreased LCD.

Table 1
Comparison of baseline characteristics among the study groups
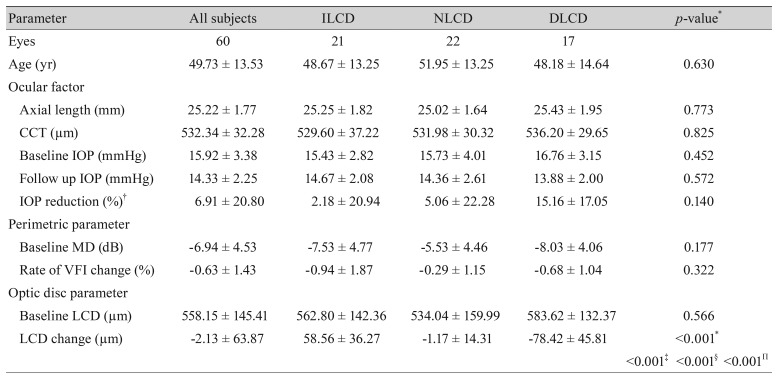
Values are presented as number or mean ± standard deviation.
LCD = lamina cribrosa depth; ILCD = increased LCD; NLCD = no LCD change; DLCD = decreased LCD; CCT = central corneal thickness; IOP = intraocular pressure; MD = mean deviation; VFI = visual field index.
*p< 0.05 considered as statistically significant; †IOP reduction = (baseline IOP − follow up IOP) / baseline IOP × 100. Intergroup ANOVA testing was performed using the mean values. If p-values were <0.05, post hoc analysis (Tukey's test) was performed; ‡p-value for the post hoc analysis of the ILCD and NLCD groups; §p-value for the post hoc analysis of the NLCD and DLCD groups; Πp-value for the post hoc analysis of the ILCD and DLCD groups.
- TOOLS
-
METRICS

-
- 4 Crossref
- 0 Scopus
- 2,269 View
- 27 Download
- Related articles



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



