 |
 |
| Korean J Ophthalmol > Volume 31(2); 2017 > Article |
Abstract
Purpose
The aim of this study was to investigate the differences in anterior segment ocular parameters in anisometropia >1 D.
Methods
This study included 202 eyes of 101 subjects ranging from 10 to 40 years of age with anisometropia of 1 D or more. The subjects were divided into groups according to anisomyopia, anisoastigmatism, and anisohypermetropia. After providing informed consent, each patient underwent a detailed ophthalmological examination including cycloplegic refraction, best-corrected visual acuity, cover test, axial length (AL) measurement using A-scan ultrasound biometer, keratometry, anterior chamber depth, and central corneal thickness measurement. For each participant, the eye with greater refractive error was compared to the fellow eye via paired t-tests. Correlations between parameters were studied using the Pearson correlation coefficient.
Results
The average age of subjects was 21.7 Ā± 9.3 years. Of 101 subjects, 31 had anisomyopia; 42 had anisohypermetropia; and 28 had anisoastigmatism. A predisposition toward greater myopia in right eyes was noted in anisomyopia (24 of 31 subjects, 77%). The inter-ocular acuity difference was significant in all three groups (p < 0.01). As the degree of anisometropia increased, there was significant positive correlation in the difference in AL in myopes (r = 0.863, p < 0.01) and hypermetropes (r = 0.669, p < 0.01) and the difference in corneal curvature in anisoastigmatism (r = 0.564, p = 0.002) and hypermetropes (r = 0.376, p = 0.014). A significant positive correlation was also present between the anterior chamber depth difference and refractive difference in hypermetropes (r = 0.359, p = 0.020).
Conclusions
This study showed that anisomyopia is correlated only with anterior chamber differences. Anisohypermetropia is correlated with AL differences as well as corneal curvature difference and anterior chamber depth difference. The amount of anisoastigmatism correlates only with corneal curvature difference.
Anisometropia, an inter-ocular difference in refraction, is an important condition as it can lead to significant visual problems including impaired stereopsis amblyopia and strabismus [1,2,3]. Although there is no defined value, an inter-ocular difference of 1 diopter (D) or more is accepted as the threshold for anisometropia by most authors [4,5,6]. Anisometropia may be broadly characterized as myopic, hypermetropic, and astigmatic. It represents unequal eye growth or a stretch within the visual system, which has presumably received the same visual input. Examination of the two eyes from the same subject allows for greater control of confounding variables like genetic and environmental influences. Therefore, anisometropia is of great use in refractive error research.
Ocular biometric and topographic parameters are important measures in different fields of ophthalmology. Corneal thickness is an important parameter for planning refractive surgeries as it guides the depth of stromal ablation [7]. Central corneal thickness (CCT) and anterior chamber depth (ACD) are used in glaucoma risk prediction [8,9]. ACD, corneal curvature (K), and axial length (AL) are required for calculating the power of intraocular lenses in various theoretical biometric formulas [10,11]. Hence, an understanding of these parameters is clinically helpful for ophthalmologists, and anisometropia provides a good model.
Refractive error results from the difference in refractive power according to the curved surface of the cornea and crystalline lens, as well as the axial changes associated with AL, ACD, lens thickness, and vitreous chamber depth [12,13]. Many prior studies have reported a correlation between the refractive error and ocular parameters. Olsen et al. [14] reported that ocular refraction is correlated with corneal power, lens power, and the AL. Wong et al. [15] reported that vitreous chamber depth was the most important determinant of refraction in adults over 40 years of age. However, the mechanism associated with the development of anisometropia has not been well-defined in most studies. Moreover, most studies have focused on myopic anisometropia and how it correlates with ocular parameters, and hypermetropic and astigmatic anisometropia and their correlations have not been well-studied. This study aimed to determine whether the eyes of anisometropic patients have different ocular parameters, such as AL, K, ACD, and CCT, and how they correlate with the anisometropia. Laterality, visual acuity and prevalence of amblyopia in different types of anisometropia were also investigated.
This prospective cross-sectional study was performed from November 2013 to April 2015. The study protocol was approved by the institutional ethics committee (IECHR/13) of University College of Medical Sciences, Delhi. Written and informed consent was obtained from adult patients and parents or legal guardians of minor patients prior to enrollment.
The study included 202 eyes of 101 subjects ranging from 10 to 40 years of age with anisometropia of 1 D or more. After informed consent, a detailed ophthalmological examination was performed including cycloplegic refraction; best-corrected visual acuity (BCVA), AL, K, ACD, and CCT measurement. To induce pupil dilatation, three drops of 0.8% tropicamide were instilled 10 minutes apart and autorefraction was performed. The mean of three successful measurements was used for analysis. The cycloplegic spherical equivalent (spherical component +1 / 2 cylinder component) was used to calculate refractive error. An inter-ocular difference of 1.00 D or more in cycloplegic spherical equivalent was considered anisometropia. Amblyopia was defined as visual acuity of 20 / 30 or worse, or a difference of two or more lines on Snellen's chart between the eyes.
Subjects with any of the following ocular pathologies were excluded from the study: corneal opacity, cataract, nystagmus, history of previous ocular surgery, retinal pathology, and those who were not sufficiently cooperative for ocular examinations.
To assess BCVA, patients were retested 3 days after cycloplegic refraction. In order to calculate and compare visual acuity, Snellen acuity was converted to logarithm of the minimal angle of resolution (logMAR). In addition, the eye with greater refractive error was also recorded to compare the laterality.
AL was measured using an A-scan biometer (APPASCAN 2000; Appasamy Associates, Chennai, India). Horizontal and vertical Ks were measured using a Bausch and Lomb keratometer (Rochester, NY, USA). ACD was measured by Haag Streit optical pachymeter using depth measuring devices I and II which were mounted on Haag Streit slit lamp (BQ900; Haag Streit, Koeniz, Switzerland). CCT was measured by ultrasound pachymeter. The average of three sets of measurements was calculated and recorded for each parameter.
For each participant, the eye with greater refractive error was compared to the fellow eye via paired t-tests using SPSS ver. 19.0 (IBM Corp., Armonk, NY, USA) with p < 0.05 considered significant. Correlations between parameters were studied using the Pearson correlation coefficient and simple linear regression.
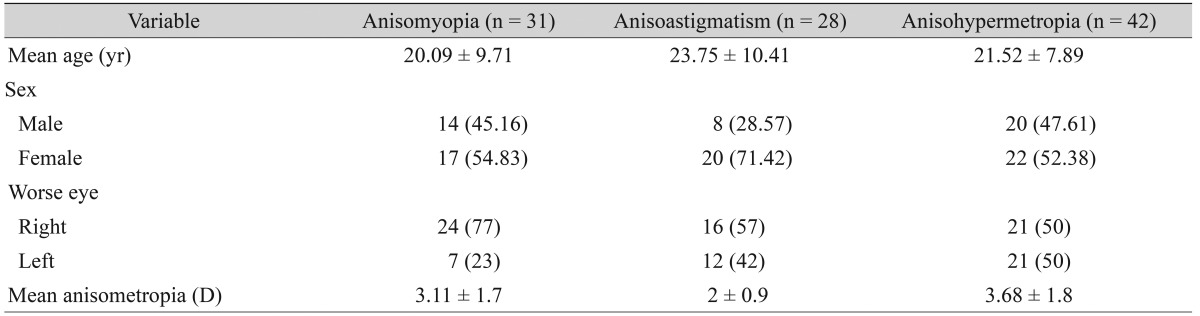
The average age of 101 subjects was 21.7 Ā± 9.3 years (range, 10 to 40 years). There were 59 females and 42 males. All subjects had anisometropia of 1 D or more, with 31 subjects having anisomyopia, 42 having anisohypermetropia, and 28 having anisoastigmatism. The demographic profile and the mean degree of anisometropia in the three groups are presented in Table 1. The male : female ratio was 2 : 3 in anisomyopic and anisohypermetropic subjects; however, there were more females (71.42%) than males (28.57%) with astigmatic anisometropia. The mean anisometropia was 3.11 Ā± 1.7 D in myopia, 2 Ā± 0.99 D in astigmatism, and 3.68 Ā± 1.85 D in hypermetropia.
In anisomyopia, the right eye was more myopic in most subjects (24 of 31, 77%). This difference in laterality was not found in the meridional and hypermetropic groups (Table 1).
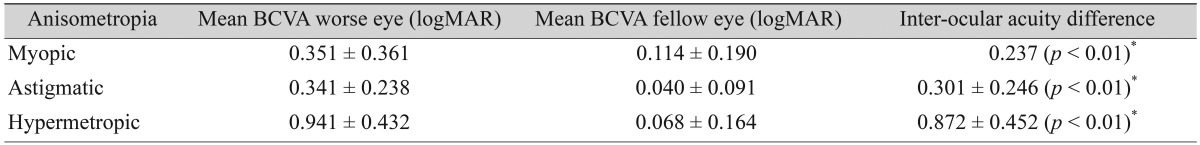
The mean BCVA (logMAR) of each eye in the three anisometropia groups is shown in Table 2. The inter-ocular acuity difference was significant in all groups (p < 0.01). Anisohypermetropes had the worst visual acuity in the affected eye when compared to anisomyopes and anisoastigmatic subjects.
The inter-ocular acuity difference was found to be linearly correlated with the degree of anisometropia (Fig. 1A-1C).
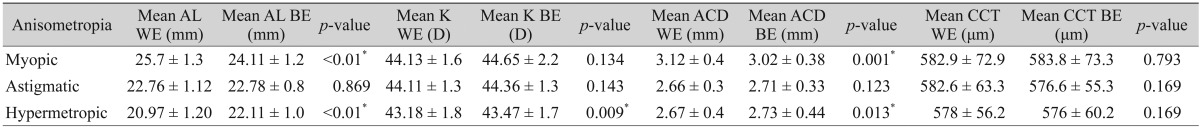
The ocular component values of AL, K, ACD, and CCT are presented in Table 3.
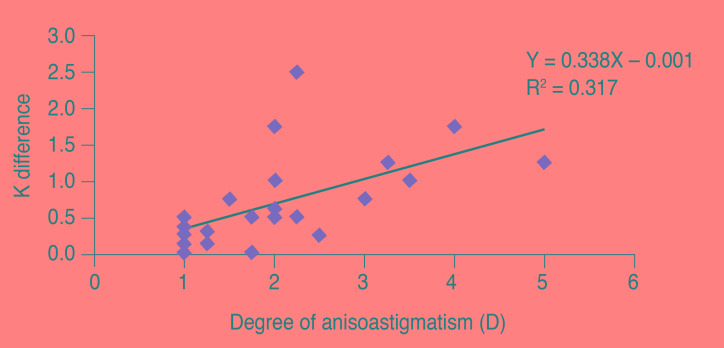
As the degree of anisometropia increased, there was a significant positive correlation in the difference in AL length in myopes (r = 0.863, p < 0.01) and hypermetropes (r = 0.669, p < 0.01) (Fig. 2A and 2B) and the difference in K in astigmatism (r = 0.564, p = 0.002) (Fig. 3) and hypermetropes (r = 0.376, p = 0.014). A significant positive correlation was also present between the ACD difference and refractive difference in hypermetropes (r = 0.359, p = 0.020).
Of 101 anisometropic subjects, 69 (68.31%) had amblyopia. Eleven of these 69 amblyopic subjects (15.9%) had myopic anisometropia; 18 (26.08%) had meridional anisometropia; and 40 (57.97%) had hypermetropic anisometropia.
In this study, we evaluated the anterior segment ocular parameters of subjects with anisometropia. In addition, we compared visual acuity, sex, laterality, and the presence of amblyopia in different types of anisometropia. We found a tendency for right eyes to have more spherical myopia when compared to left eyes (77% of anisomyopic subjects had greater refractive error in the right eye). This difference in laterality was not found in hypermetropic and astigmatic anisometropia. This finding is similar to that of a study in Hong Kong children, which reported a tendency for right eye laterality in myopic subjects [16]. Similarly, right eye laterality in myopic subjects with anisometropia >2.5 D was also reported by Linke et al. [17].
In this study, hypermetropic anisometropes had the worst BCVA, followed by astigmatic and myopic subjects (Table 2). The inter-ocular acuity difference between fellow eyes was found to be linearly correlated with the amount of anisometropia (Fig. 1A-1C). The mean inter-ocular acuity difference was statistically significant in all three types of anisometropia (p < 0.01). Thus, visual acuity appears to deteriorate with increasing anisometropia, and hypermetropic anisometropes were the most severely affected.
Similar results were reported by Weakley [18] in their study of visual function in anisometropes. They found a significant increase in the inter-ocular acuity difference in the presence of myopia, hypermetropia, and astigmatic anisometropia. They also found hypermetropic subjects to have the worst acuity. Studies by Dobson et al. [19], Levi et al. [20], and Chen et al. [21] reported similar associations between poor visual acuity and anisometropia.
When comparing the mean ocular parameters in the three groups, we found a significant difference in AL and ACD in both anisomyopia and anisohypermetropia. The more myopic eyes of anisomyopic patients were significantly longer (p < 0.01), while the more hypermetropic eyes were significantly shorter (p < 0.01) than fellow eyes. The anterior chamber of the more myopic eyes in cases of anisomyopia was significantly deeper than in fellow eyes (p = 0.001). However, when we tried to correlate the refractive difference with ACD difference between pair of eyes in anisomyopia, we did not find any significant correlation (r = ā0.48, p = 0.799). These results are similar to those of previous studies [15,22] which reported no correlation of anisomyopia with ACD. Both these studies, however, found a positive correlation between anisomyopia and vitreous chamber depth, measured indirectly by A-scan, which we were not able to calculate in our study. Thus, our results support the theory of deeper ACD in myopia [22,23]. However, we found that ACD in myopes does not vary proportionately with the amount of refractive error. Our results further support the theory that the difference in refractive error between the eyes of anisomyopic patients is predominantly axial in nature and is determined by the difference in length of the posterior segment.
Our findings were in agreement with studies on anisomyopia by Tekin et al. [24], Kim et al. [25], and Logan et al. [26] who found more myopic eyes to be longer and the degree of anisometropia to be correlated with AL difference. Thus, our study supports the hypothesis that axial elongation, rather than K, is the major contributing factor in myopia [27,28]. Our study also found a linear correlation between axial difference and refractive difference between pairs of eyes.
Anisohypermetropia, on the other hand, was found to be multifactorial, as there was a significant correlation between the refractive difference and AL, K, and ACD difference between fellow eyes. Like myopia, hypermetropia is also axial in nature; however, K plays a significant role in hypermetropia unlike in myopia. Similar findings were reported by Strang et al. [29] in a study of hypermetropic subjects. They found a weak but statistically significant correlation between mean K and refractive error, with corneas getting flatter as hypermetropia increased. Similar to our study, Demircan et al. [30] found a statistically significant difference in K, ACD and AL between fellow eyes in hyperopic anisometropic amblyopia. However, Yuksel et al. [31] found no significant difference among anterior segment ocular parameters in hyperopic anisometropic amblyopes, although their study included normal subjects as controls. This may account for the differences in the results.
Anisoastigmatism correlated with K difference only (r = 0.564, p = 0.002). A similar correlation was reported by Huynh et al. [5], who found a correlation between the refractive difference and K difference between pairs of eyes.
In short, our study showed that while anisomyopia is correlated with AL difference only (r = 0.863, p < 0.01), anisohypermetropia is correlated with AL difference (r = 0.669, p <0.01) as well as K difference (r = 0.376, p = 0.014) and AC depth difference (r = 0.359, p = 0.020). The amount of anisoastigmatism correlated only with K difference (r = 0.564, p = 0.002). Anisometropic eyes provide us with the chance to understand biometric change in eyeballs with different refractive statuses in the same person. Such information is helpful for calculating the intraocular lens power in cataract surgery, planning refractive surgery and understanding the risk factors for glaucoma in eyes of different refractive power.
A high percentage of anisometropic subjects (68.31%) in our study had amblyopia. The reported prevalence of amblyopia in anisometropia varied from 25% to 60% in previous studies [32]. The higher frequency of amblyopia in our study may be due to selection bias, as ours was a small hospital-based study, while others were population-based. Moreover, our study included more patients with hypermetropic anisometropia (41%) and higher degrees (>3 D) of anisometropia (47.5% of patients), both of which have greater odds of developing amblyopia.
Our study was limited by a small number of subjects. Furthermore, a hospital-based population is more likely to have severe disabilities. We did not have a control group of normal children and adults. A similar population-based study on a larger group of subjects is needed to confirm these results. Our findings suggest that anisometropia has an impact on anterior segment ocular parameters, but this will need to be confirmed by further histopathological studies.
Notes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
REFERENCES
2. Weakley DR Jr. The association between nonstrabismic anisometropia, amblyopia, and subnormal binocularity. Ophthalmology 2001;108:163-171.


3. Weakley DR Jr, Birch E, Kip K. The role of anisometropia in the development of accommodative esotropia. J AAPOS 2001;5:153-157.


4. O'Donoghue L, McClelland JF, Logan NS, et al. Profile of anisometropia and aniso-astigmatism in children: prevalence and association with age, ocular biometric measures, and refractive status. Invest Ophthalmol Vis Sci 2013;54:602-608.


5. Huynh SC, Wang XY, Ip J, et al. Prevalence and associations of anisometropia and aniso-astigmatism in a population based sample of 6 year old children. Br J Ophthalmol 2006;90:597-601.



6. Vincent SJ, Collins MJ, Read SA, Carney LG. Myopic anisometropia: ocular characteristics and aetiological considerations. Clin Exp Optom 2014;97:291-307.


7. Randleman JB. Post-laser in-situ keratomileusis ectasia: current understanding and future directions. Curr Opin Ophthalmol 2006;17:406-412.


8. Devereux JG, Foster PJ, Baasanhu J, et al. Anterior chamber depth measurement as a screening tool for primary angle-closure glaucoma in an East Asian population. Arch Ophthalmol 2000;118:257-263.


9. Congdon NG, Youlin Q, Quigley H, et al. Biometry and primary angle-closure glaucoma among Chinese, white, and black populations. Ophthalmology 1997;104:1489-1495.


10. Olsen T. Sources of error in intraocular lens power calculation. J Cataract Refract Surg 1992;18:125-129.


11. Holladay JT. Standardizing constants for ultrasonic biometry, keratometry, and intraocular lens power calculations. J Cataract Refract Surg 1997;23:1356-1370.


12. In: Benjamin WJ, Borish IM, Borish's clinical refraction. Philadelphia: W. B. Saunders; 1998. p. 2-17.
13. Troilo D. Neonatal eye growth and emmetropisation: a literature review. Eye (Lond) 1992;6(Pt 2):154-160.


14. Olsen T, Arnarsson A, Sasaki H, et al. On the ocular refractive components: the Reykjavik Eye Study. Acta Ophthalmol Scand 2007;85:361-366.


15. Wong TY, Foster PJ, Ng TP, et al. Variations in ocular biometry in an adult Chinese population in Singapore: the Tanjong Pagar Survey. Invest Ophthalmol Vis Sci 2001;42:73-80.

16. Goldschmidt E, Lyhne N, Lam CS. Ocular anisometropia and laterality. Acta Ophthalmol Scand 2004;82:175-178.


17. Linke SJ, Druchkiv V, Steinberg J, et al. Eye laterality: a comprehensive analysis in refractive surgery candidates. Acta Ophthalmol 2013;91:e363-e368.


18. Weakley DR. The association between anisometropia, amblyopia, and binocularity in the absence of strabismus. Trans Am Ophthalmol Soc 1999;97:987-1021.


19. Dobson V, Miller JM, Clifford-Donaldson CE, Harvey EM. Associations between anisometropia, amblyopia, and reduced stereoacuity in a school-aged population with a high prevalence of astigmatism. Invest Ophthalmol Vis Sci 2008;49:4427-4436.



21. Chen BB, Song FW, Sun ZH, Yang Y. Anisometropia magnitude and visual deficits in previously untreated anisometropic amblyopia. Int J Ophthalmol 2013;6:606-610.


22. Tian Y, Tarrant J, Wildsoet CF. Optical and biometric characteristics of anisomyopia in human adults. Ophthalmic Physiol Opt 2011;31:540-549.



23. In: Curtin BJ, The myopias: basic science and clinical management. Philadelphia: Harper and Row; 1985. p. 277-385.
24. Tekin K, Cankurtaran V, Inanc M, et al. Effect of myopic anisometropia on anterior and posterior ocular segment parameters. Int Ophthalmol 2017;37:377-384.


25. Kim SY, Cho SY, Yang JW, et al. The correlation of differences in the ocular component values with the degree of myopic anisometropia. Korean J Ophthalmol 2013;27:44-47.



26. Logan NS, Gilmartin B, Wildsoet CF, Dunne MC. Posterior retinal contour in adult human anisomyopia. Invest Ophthalmol Vis Sci 2004;45:2152-2162.


27. Goss DA, Erickson P. Meridional corneal components of myopia progression in young adults and children. Am J Optom Physiol Opt 1987;64:475-481.


28. Shih YF, Chiang TH, Lin LL. Lens thickness changes among schoolchildren in Taiwan. Invest Ophthalmol Vis Sci 2009;50:2637-2644.


29. Strang NC, Schmid KL, Carney LG. Hyperopia is predominantly axial in nature. Curr Eye Res 1998;17:380-383.


30. Demircan S, Gokce G, Yuvaci I, et al. The assessment of anterior and posterior ocular structures in hyperopic anisometropic amblyopia. Med Sci Monit 2015;21:1181-1188.



Fig.Ā 1
(A) The correlation between the degree of anisomyopia and inter-ocular acuity difference (IAD). (B) The correlation between anisoastigmatism and IAD. (C) The correlation between anisohypermetropia and IAD. logMAR = logarithm of the minimal angle of resolution; D = diopter.
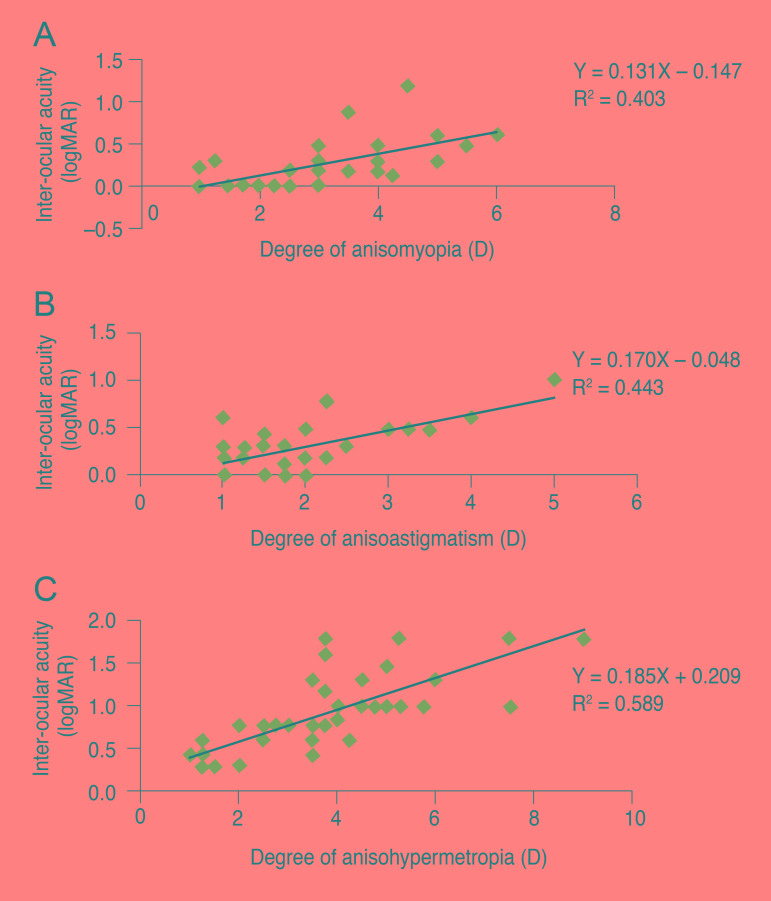
Fig.Ā 2
The linear correlation between the difference in axial length and degree of anisometropia between fellow eyes anisomyopia (A) and anisohypermetropia (B).

Fig.Ā 3
The correlation between corneal curvature (K) difference and refractive difference between fellow eyes in anisoastigmatism.
- TOOLS
-
METRICS

-
- 11 Crossref
- 0 Scopus
- 2,141 View
- 11 Download
- Related articles
-
Evaluation of Anterior Segment Parameters in Obesity2015 August;29(4)
A retrospective study of 1,073 cases of intraocular lens implantation.1987 June;1(1)



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



