The prevalence of myopia has profoundly increased, and laser refractive surgery is a popular procedure for safe and effective myopic correction. Many studies have shown good visual acuity and stable refractive outcomes after myopic laser refractive surgery. Laser in situ keratomileusis (LASIK) is predominantly performed, due to its rapid visual outcome and little pain. In contrast, laser-assisted subepithelial keratectomy (LASEK) is preferred in cases with thin corneas and high myopia. However, the tendency toward myopia after laser refractive surgery has been reported frequently and is difficult for ophthalmologists to address.
Regression is the tendency of an eye to return to its original refraction and is dependent on the preoperative myopia and the amount of correction [
1,
2]. A previous study of LASIK reported that the myopic regression for moderate to high myopia was -1.66 ± 2.15 diopters (D) over 15 years, indicating a regression rate of -0.11 D per year [
3]. LASIK performed with a mechanical microkeratome may be a factor affecting myopia regressions due to uneven flap thickness [
4]. Also, a considerable regression of high myopia over time after laser refractive surgery is a well-known phenomenon [
5,
6]. Recently, LASEK has been used for the correction of high myopia and an increased incidence of regressions has been reported in LASEK patients.
Laser refractive surgery is frequently performed in moderate myopia, and most patients demonstrate successful outcomes; however, there is little knowledge about the general changes in visual acuity and the refractive tendencies of patients without postoperative problems. Most studies have tried to identify risk factors in specific cases, such as those involving ectasia or retreatment, rather than in general cases. Patients may represent different degrees of regressions toward myopia, and the correlation between the amount of myopic regression and the visual acuity is not clear. Some patients are satisfied with a certain amount of myopia, but other patients complain of discomfort despite the existence of only a small degree of myopia. Patient's tolerances are variable, and thus, determining the need for retreatment is very difficult. It is essential to evaluate the eyes with myopic regression in patients who did not require retreatment for a long period.
The purpose of this study was to identify general refractive changes and to compare significant risk factors for regression between LASIK and LASEK for moderate myopia during a 10-year follow-up period.
Materials and Methods
Retrospective data were collected from February 1998 to February 2003 at Seoul St. Mary's Hospital. The study protocol followed the guidelines of the Declaration of Helsinki and was approved by the hospital's institutional review board. The charts of all patients who had LASIK or LASEK performed by the same surgeon (CKJ) were reviewed. Among these patients, we included those that had a preoperative spherical error of less than -10 D and a cylinder error of less than -3 D and those that returned for examinations annually, specifically at 6 months and 1, 2, 5, 7, and 10 years after surgery.
All surgery targeted distance vision. All patients were counseled on refractive surgery and provided informed consent before the surgery. The patients were examined with ophthalmologic tests preoperatively and at 6 months and 1, 2, 5, 7, and 10 years after surgery. The uncorrected visual acuity (UCVA), best spectacle corrected visual acuity (BSCVA), manifest refraction, slit lamp examination, intraocular pressure (IOP), ultrasound pachymetry, and corneal topography (Orbscan II; Bausch & Lomb, Rochester, NY, USA) were examined.
The inclusion criteria were the myopic refractive error and normal preoperative topography. The exclusion criteria included ocular pathology, retinal disorders, hyperopia (positive spherical equivalent), and insufficient follow-up. Patients with ocular or general medical histories such as diabetes were also excluded. All patients had stable refraction for at least 1 year before surgery. When patients were evaluated after surgery, we excluded those with topographic signs suggesting corneal instability, those who needed retreatment, and those who had corneal haze or other complications.
We defined the regression group as patients having myopia of more than 1 D at 10 years postoperatively and the non-regression group as patients having myopia less than 1 D at 10 years postoperatively following LASIK or LASEK procedures.
Surgical technique
An experienced surgeon performed all of the procedures. The preoperative regimen was similar for all patients, and anesthesia consisted of topical eyedrops of proparacaine (Alcaine; Alcon-Couvreur, Puur, Belgium). LASIK and LASEK were performed using a VISX S4 excimer laser system (VISX Inc., Santa Clara, CA, USA). Refractive surgery was performed with a 6.5-mm optical zone with no wavefront-guided treatments.
In LASIK-treated eyes, a microkeratome (Moria, Antony, France) was applied to create a nasal-hinged flap using the manufacturer's recommended parameters. Excimer laser stromal ablation was performed after the flap was reflected nasally. Immediately after ablation, the stromal bed was irrigated with a balanced salt solution, and the flap was positioned. In eyes treated with LASEK, the corneal epithelium was removed with a 20% alcohol solution for 30 seconds and then irrigated with a balanced salt solution. Laser surface ablation was performed using an excimer laser based on the surgeon's criteria.
After LASIK and LASEK surgeries, drops of fluoroquinolone antibiotics and fluorometholone 0.1% were instilled into the lower fornix. Topical antibiotic and corticosteroid agents were applied four times daily for a minimum of four weeks, and gradual tapering was applied based on refraction, IOP, and corneal status.
Statistical analysis
The visual outcomes evaluated included the efficacy index (postoperative UCVA / preoperative BSCVA) and the safety index (postoperative BSCVA / preoperative BSCVA). The refractive outcome was evaluated as predictability, which was defined as the percentage of the eye within 1.0 D in a spherical equivalent (SE).
All data are expressed as the means ± standard deviations. Statistical analyses were performed using SPSS ver. 17.0 (SPSS Inc., Chicago, IL, USA). Demographic data were compared using chi-square tests. Paired t-tests were used to compare preoperative and postoperative visual and refractive outcomes. Comparisons between LASIK and LASEK were performed using unpaired t-tests. The p-values less than 0.05 were considered statistically significant.
Results
Laser refractive surgery was performed on a total of 487 eyes between June 1, 1998 and December 31, 2003. According to above inclusion criteria, 425 patients were excluded: 203 were lost to follow-up; 133 were not satisfied the inclusion of refraction (hyperopia, low myopia, high myopia, and severe astigmatism); 46 had corneal abnormalities upon follow-up (topographic abnormality, corneal haze, ectasia, and infection); and 43 had systemic diseases (diabetes, autoimmune disease, and cancer). After these exclusions, 62 eyes were evaluated in annual follow-ups and were included in this study. Thirty-six eyes had LASIK, and 26 eyes had LASEK. The preoperative SEs ranged from -3.5 to -8.625 D (mean ± SE, -5.73 ± 2.76 D) in the LASIK group and from -3 to -8.625 D (mean ± SE, -5.45 ± 1.84 D) in the LASEK group. The patient's preoperative data are shown in
Table 1. Age, refraction, and visual acuity were similar in the LASIK and LASEK groups. Central corneal thickness (CCT) and IOP were significantly different in the two groups.
Efficacy
UCVA showed significant improvements at all visits in both groups (p < 0.001). The efficacy index (postoperative UCVA / preoperative BSCVA) of LASEK was consistently higher than that of LASIK in moderate myopia. Between the LASIK and LASEK groups, significant differences were apparent at 2, 5, and 10 years after surgery.
Safety
Safety indices (postoperative BSCVA / preoperative BSCVA) were above 0.90 in both groups. The safety index of LASEK was higher than that of LASIK, but the differences in the safety indices were not significant between the groups during follow-up.
Table 2 shows the safety and efficacy indices of the LASIK and LASEK patients.
Predictability
The predictability of the refractive surgery is shown in
Fig. 1A and 1B. In the LASIK group, postoperative mean refractive spherical equivalent (MRSE) within ±1.00 D was 93.3% after 6 months and 68.3% after 10 years. In the LASEK group, postoperative MRSE within ±1.00 D was 82.5% after 6 months and 77.5% after 10 years. The predictability was higher in the LASIK group than the LASEK group at 6 months after surgery. However, 10 years after surgery, the LASEK group showed superior predictability.
Refractive change
The MRSEs were significantly improved for all follow-up periods. The difference between the SE 6 months postoperatively and at later periods was statistically significant at 5, 7, and 10 years in both groups (LASIK,
p = 0.036,
p = 0.003, and
p = <0.001, respectively; LASEK,
p = 0.006,
p = 0.002, and
p = 0.001, respectively). The mean SEs were comparable between the LASIK and LASEK groups at all follow-up intervals in myopic eyes (
p > 0.05). Myopic change was observed over time in both groups, even years after the initial surgical procedure.
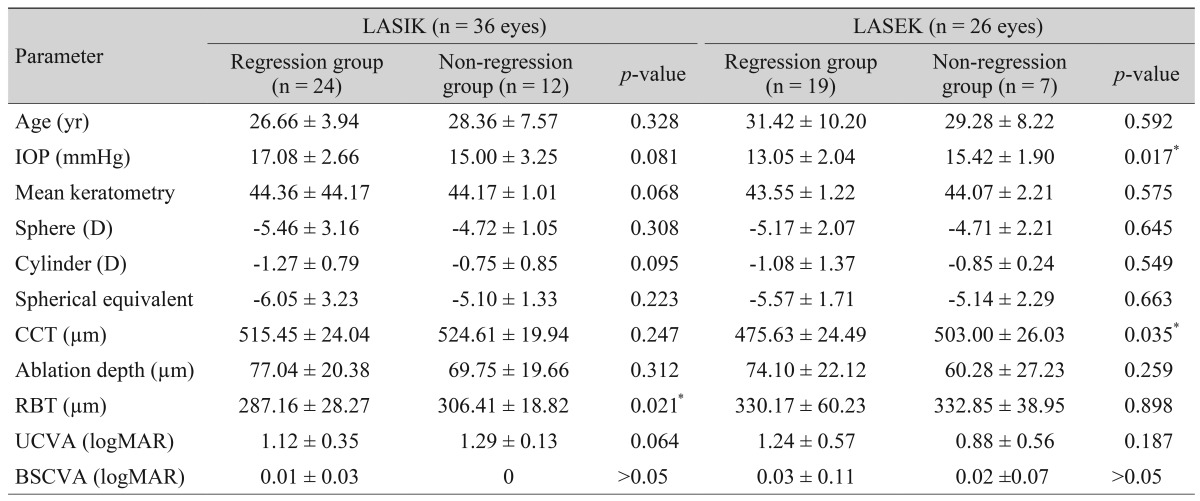
Fig. 2A and 2B shows the MRSE over time by surgery (LASIK or LASEK) and by degree of myopia 10 years postoperatively in all eyes over the duration of the study. The regression group (myopia of more than 1 D 10 years postoperatively) showed an early myopic tendency 1 year after surgery in the LASIK group and 6 months to 10 years after surgery, except at 5 years, in the LASEK group as compared with those in the non-regression group (myopia less than 1 D 10 years post-operatively). Preoperative characteristics of the regression and non-regression groups are shown in
Table 3.
Central corneal thickness
Fig. 3A and 3B shows the mean CCT over time by surgery (LASIK or LASEK) and by degree of myopia 10 years postoperatively. There was an increase in CCT after myopic LASIK and LASEK in both the regression and non-regression groups. In comparison with the CCT at 6 months postoperatively, a significant increase was noted after 5, 7, and 10 years in both LASIK and LASEK patients (LASIK,
p < 0.001,
p < 0.001, and
p < 0.001, respectively; LASEK,
p = 0.01,
p < 0.001, and
p < 0.001, respectively). There were no significant differences between the regression and non-regression groups in either LASIK or LASEK patients.
Discussion
LASIK is the most popular refractive surgery, whereas LASEK is commonly used in patients with thin corneas or dry eyes [
7,
8]. This study evaluated the visual and refractive outcomes of LASIK compared to LASEK. We focused on the differences and changes between patients having myopia of more than 1 D (regression group) and those having myopia of less than 1 D (non-regression group) 10 years postoperatively. Myopia of more than 1 D was seen in 76.6% of the LASIK group and 73.0% of the LASEK group 10 years after surgery. The mean myopic change throughout the 10-year follow-up period was -1.09 D in LASIK patients and -1.34 D in LASEK patients.
Previous studies showed variable results regarding the efficacies of LASIK and LASEK for the correction of myopia. Tobaigy et al. [
9] and Scerrati [
10] suggested that visual acuity and refractive outcomes were better in LASEK than in LASIK. Kim et al. [
11] concluded that LASIK surgery was superior to LASEK for high myopia. We retrospectively reviewed 10-year follow-up charts and found that LASIK and LASEK had similar efficacies for moderate myopia within 2 years, with LASEK producing significantly superior efficacy 4 years postoperatively. Eyes treated with LASIK had a lower efficacy index of less than 0.8 at 5 years postoperatively and might show greater myopic regression. Safety indices were similar for LASIK and LASEK in our study.
Refractive changes after surgery were variable, although we aimed to correct for refractive error [
12]. A myopic shift after myopic laser refractive surgery is well known and is thought to result from undercorrection or regression [
1,
13]. The causes of the regression can be divided into corneal and non-corneal causes. Non-corneal causes include inaccurate preoperative refraction and inadequate laser energy delivery [
1,
14]. Corneal causes include biomechanical changes of the cornea after the keratectomy. Manifest SE was increased for up to 10 years after refractive surgery to correct myopia, and this tendency was shown over time in both groups, even years after the initial surgical procedure. The mean myopic change throughout the 10-year follow-up period was -1.09 D in LASIK patients and -1.34 D in LASEK patients. Our results showed more myopic change than did previous studies [
15,
16,
17]. Because myopic regression is difficult to define with a specific value, we defined the regression group as patients having myopia of more than 1 D. According to Hirsch [
18], a myopia of 1 D is approximately the 6 / 18 level of visual acuity, which defines the limitation of vision impairment according to the World Health Organization [
19]. Between the regression and non-regression groups, the mean SEs were significantly different 1 year after surgery in the LASIK group and 6 months after surgery in the LASEK group.
We speculated that long-term myopic status was correlated with preoperative refraction, CCT, and ablation profile. Compared with the non-regression group, the regression group had significantly lower residual bed thickness (RBT) preoperatively in the LASIK patients (
p = 0.021) and lower CCT in the LASEK patients (
p = 0.035). Wang et al. [
20] reported that an RBT below 250 µm increased the risk of ectasia after LASIK [
20]. Erie et al. [
21] also found that an RBT of 250 µm does not safely prevent the development of keratectasia. Pan et al. [
13] found that refractive regression after LASIK is mainly caused by corneal protrusion rather than postoperative central corneal thickening or RBT. Our study indicated that myopic regression after LASIK was correlated with a low RBT preoperatively. In LASEK, patients with myopic regression had a mean preoperative CCT thinner than 500 µm. Some studies reported that surface ablation had stable visual and refractive outcomes in corneas thinner than 500 µm at long-term follow-up [
22]. Ectasia after LASEK is rare and was correlated with abnormal topography and CCT preoperatively [
20,
23]. Our results showed that regression in LASEK patients was correlated with low CCT and IOP, but not with RBT.
Pan et al. [
13] reported a progressive increase in CCT in the eyes in the regression group than in the non-regression group after LASIK in high myopic patients. Some studies also reported that the eyes with refractive regression after LASIK and photorefractive keratectomy showed a progressive increase in CCT [
1,
24]. In our study, postoperative CCT increased significantly with time. CCT was significantly smaller at 6 months postoperatively than after 5, 7, or 10 years postoperatively in both surgeries. Some studies reported that an increase in central epithelial thickness after refractive surgery is related to myopic regression [
25,
26,
27], but we did not detect a correlation between myopic regression and change in CCT. Our results are consistent with previous studies in that total corneal thickening may not completely explain the refractive change after laser refractive surgery [
28]. It has been reported that the corneal epithelium has a tendency to restore its original contour and could be a nonuniform thickness during the wound healing process, leading to the development of refractive power [
26,
27,
29]. Lohmann and Guell [
27] reported that a difference in the epithelium of approximately 10 µm caused a refractive change of 1 D. Further study is needed to compare the epithelial thicknesses between the regression and non-regression groups.
This study was a retrospective comparative case series, so we evaluated only the patients that came to our center regularly. The small number of patients was a limitation in this study, and a greater length of follow-up is needed to understand the natural course of laser refractive surgery. Significant differences in CCT, RBT, and IOP were observed between the patients who had LASIK and those who had LASEK. Still, preoperative data were well controlled between the regression and non-regression patients. Ten years ago, we performed blade-assisted flap creation rather than femtosecond laser-assisted flap creation and could not consider wave-front guided laser surgery.
In conclusion, our study with 10 years of follow-up demonstrated that both LASIK and LASEK performed to correct moderate myopia showed progressive myopic shifts and corneal thickening. Significant differences in SE between the regression and non-regression groups began 1 year postoperatively in the LASIK group and 6 months postoperatively in the LASEK group. A larger sample size and more variable degrees of myopia are needed to support our study.
Conflicts of interest
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
REFERENCES
1. Chayet AS, Assil KK, Montes M, et al. Regression and its mechanisms after laser in situ keratomileusis in moderate and high myopia.
Ophthalmology 1998;105:1194-1199.


2. Pang G, Zhan S, Li Y, et al. Myopic regression after photorefractive keratectomy.
Zhonghua Yan Ke Za Zhi 1998;34:451-453.

3. Alio JL, Soria F, Abbouda A, Pena-Garcia P. Laser in situ keratomileusis for -6.00 to -18.00 diopters of myopia and up to -5.00 diopters of astigmatism: 15-year follow-up.
J Cataract Refract Surg 2015;41:33-40.


4. Lin MY, Chang DC, Hsu WM, Wang IJ. Cox proportional hazards model of myopic regression for laser in situ keratomileusis flap creation with a femtosecond laser and with a mechanical microkeratome.
J Cataract Refract Surg 2012;38:992-999.


5. Kim G, Christiansen SM, Moshirfar M. Change in keratometry after myopic laser in situ keratomileusis and photorefractive keratectomy.
J Cataract Refract Surg 2014;40:564-574.


6. Seiler T. Current evaluation of myopia correction with the excimer laser.
Ophthalmologe 1995;92:379-384.

7. Sutton GL, Kim P. Laser in situ keratomileusis in 2010: a review.
Clin Experiment Ophthalmol 2010;38:192-210.


8. Hammond MD, Madigan WP Jr, Bower KS. Refractive surgery in the United States Army, 2000-2003.
Ophthalmology 2005;112:184-190.


9. Tobaigy FM, Ghanem RC, Sayegh RR, et al. A control-matched comparison of laser epithelial keratomileusis and laser in situ keratomileusis for low to moderate myopia.
Am J Ophthalmol 2006;142:901-908.


10. Scerrati E. Laser in situ keratomileusis vs. laser epithelial keratomileusis (LASIK vs. LASEK).
J Refract Surg 2001;17(2 Suppl):S219-S221.


11. Kim JK, Kim SS, Lee HK, et al. Laser in situ keratomileusis versus laser-assisted subepithelial keratectomy for the correction of high myopia.
J Cataract Refract Surg 2004;30:1405-1411.


12. Liu HQ, Shi JP, Ma CR, et al. Multifactor analysis of the reasons causing undercorrection after laser in situ keratomileusis.
Zhonghua Liu Xing Bing Xue Za Zhi 2003;24:307-309.

13. Pan Q, Gu YS, Wang J, et al. Differences between regressive eyes and non-regressive eyes after LASIK for myopia in the time course of corneal changes assessed with the Orbscan.
Ophthalmologica 2004;218:96-101.


14. Netto MV, Mohan RR, Ambrosio R Jr, et al. Wound healing in the cornea: a review of refractive surgery complications and new prospects for therapy.
Cornea 2005;24:509-522.


15. O'Connor J, O'Keeffe M, Condon PI. Twelve-year follow-up of photorefractive keratectomy for low to moderate myopia.
J Refract Surg 2006;22:871-877.


16. O'Doherty M, O'Keeffe M, Kelleher C. Five year follow up of laser in situ keratomileusis for all levels of myopia.
Br J Ophthalmol 2006;90:20-23.



17. Reinstein DZ, Srivannaboon S, Gobbe M, et al. Epithelial thickness profile changes induced by myopic LASIK as measured by Artemis very high-frequency digital ultrasound.
J Refract Surg 2009;25:444-450.



18. Hirsch MJ. Relation of visual acuity to myopia.
Arch Ophthal 1945;34:418-421.


19. Wood PH. Appreciating the consequences of disease: the international classification of impairments, disabilities, and handicaps.
WHO Chron 1980;34:376-380.

20. Wang Z, Chen J, Yang B. Posterior corneal surface topographic changes after laser in situ keratomileusis are related to residual corneal bed thickness.
Ophthalmology 1999;106:406-409.


21. Erie JC, Patel SV, McLaren JW, et al. Effect of myopic laser in situ keratomileusis on epithelial and stromal thickness: a confocal microscopy study.
Ophthalmology 2002;109:1447-1452.


22. Dirani M, Couper T, Yau J, et al. Long-term refractive outcomes and stability after excimer laser surgery for myopia.
J Cataract Refract Surg 2010;36:1709-1717.


23. de Benito-Llopis L, Alio JL, Ortiz D, et al. Ten-year follow-up of excimer laser surface ablation for myopia in thin corneas.
Am J Ophthalmol 2009;147:768-773.e1-2.


24. Randleman JB, Caster AI, Banning CS, Stulting RD. Corneal ectasia after photorefractive keratectomy.
J Cataract Refract Surg 2006;32:1395-1398.


25. Reinstein DZ, Ameline B, Puech M, et al. VHF digital ultrasound three-dimensional scanning in the diagnosis of myopic regression after corneal refractive surgery.
J Refract Surg 2005;21:480-484.


26. Spadea L, Fasciani R, Necozione S, Balestrazzi E. Role of the corneal epithelium in refractive changes following laser in situ keratomileusis for high myopia.
J Refract Surg 2000;16:133-139.


27. Lohmann CP, Guell JL. Regression after LASIK for the treatment of myopia: the role of the corneal epithelium.
Semin Ophthalmol 1998;13:79-82.


28. Magallanes R, Shah S, Zadok D, et al. Stability after laser in situ keratomileusis in moderately and extremely myopic eyes.
J Cataract Refract Surg 2001;27:1007-1012.


29. Dierick HG, Missotten L. Is the corneal contour influenced by a tension in the superficial epithelial cells? A new hypothesis?
Refract Corneal Surg 1992;8:54-59.

Fig. 1
Predictability for 10 years after (A) laser in situ keratomileusis (n = 36 eyes) and (B) laser-assisted subepithelial keratectomy (n = 26 eyes) surgeries. D = diopter.
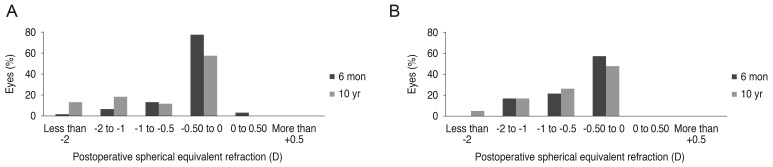
Fig. 2
Changes in mean spherical equivalents by surgery (A, laser in situ keratomileusis; B, laser-assisted subepithelial keratectomy) and by degree of myopia 10 years postoperatively.
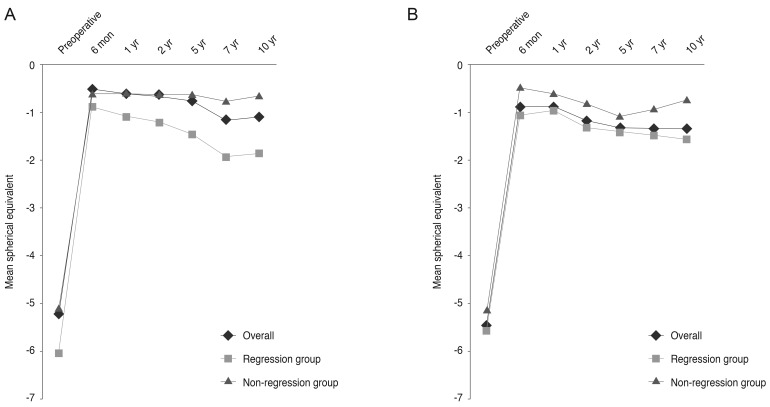
Fig. 3
Changes in mean central corneal thickness (CCT) by surgery (A, laser in situ keratomileusis; B, laser-assisted subepithelial keratectomy) and by degree of myopia 10 years postoperatively.

Table 1
Preoperative data of 62 eyes treated with laser refractive surgery that were followed for up to 10 years after surgery
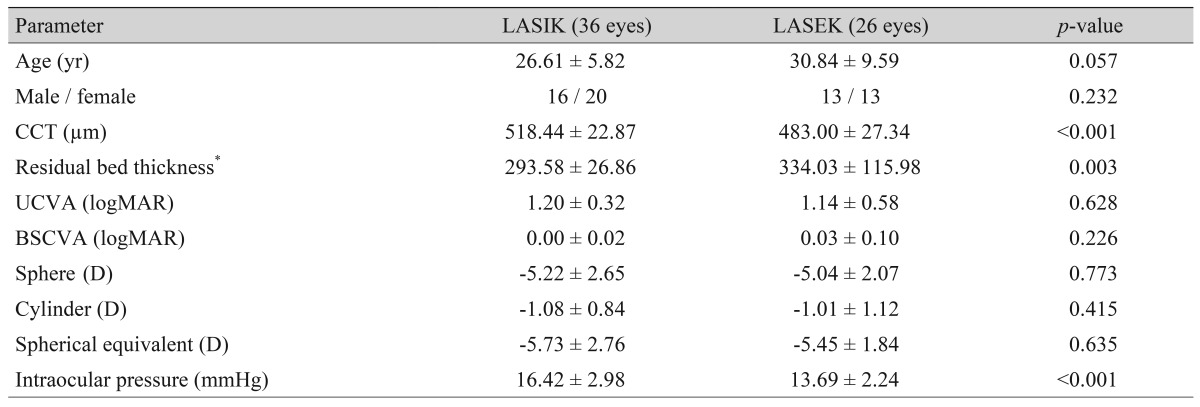
Table 2
Efficacy and safety indices for 10 years after LASIK and LASEK
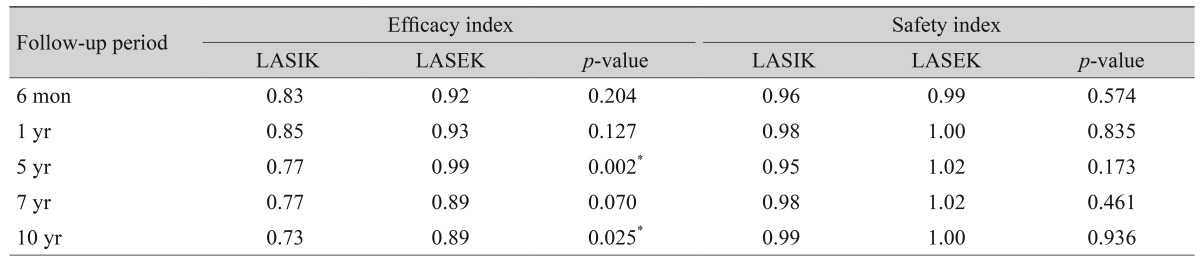
Table 3
Preoperative characteristics of regression and non-regression groups after laser refractive surgery












 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



