 |
 |
| Korean J Ophthalmol > Volume 30(5); 2016 > Article |
Abstract
Purpose
To investigate the possible associations of neutrophil-to-lymphocyte ratio (NLR) and high sensitivity C-reactive protein (hs-CRP) level with age-related macular degeneration (ARMD).
Methods
Patients were divided to three groups of 40 patients with non-neovascular ARMD (group 1), 40 patients with neovascular ARMD (group 2), and 40 healthy control subjects (group 3). The neutrophil and lymphocyte counts were evaluated using an ABX Pentra DF120/USA biochemical analyzer, and hs-CRP levels were measured using a Beckman Coulter Immage 800. The NLR was measured by dividing neutrophil count by lymphocyte count.
Results
The patients in group 2 were older and more often diabetic than the patients in groups 1 and 3 (p < 0.001 and p < 0.001, respectively). The NLR level was 1.65 ± 0.71 in group 1, 1.98 ± 0.84 in group 2, and 1.46 ± 0.44 in group 3. The hs-CRP value was 1.98 ± 0.251 mg/L in group 1, 3.242 ± 0.211 mg/L in group 2, and 1.145 ± 0.193 mg/L in group 3. Both NLR and hs-CRP values were significantly higher in group 2 compared to group 3 (p = 0.002 and p = 0.002, respectively). In multivariate analysis, NLR remained an independent predictor of neovascular ARMD (odds ratio, 3.882; 95% confidence interval, 1.574 to 9.576; p = 0.003) together with age (p < 0.001), diabetes mellitus (p = 0.041), and hs-CRP (p = 0.018).
Age-related macular degeneration (ARMD) is a one of the leading causes of blindness worldwide. ARMD is caused by the accumulation of drusen in the macula, resulting in serious vision impairment [1]. There are two subgroups of ARMD: non-neovascular ARMD and neovascular ARMD. Non-neovascular ARMD is characterized by the presence of drusen in the macula, with or without geographic atrophy. Neovascular ARMD is characterized by choroidal neovascularization and/or retinal pigment epithelium detachment in the macular region. Although neovascular ARMD represents only 10% of cases, it is responsible for almost 90% of the severe vision loss due to ARMD. Risk factors of ARMD include age, race, genetics, cigarette smoking, sunlight exposure, obesity, hypertension, and hypercholesterolemia [1]. Vascular and oxidative stress theories have been implicated for development of choroidal neovascularization in neovascular ARMD [2]. The roles of inflammatory mediators and cells as immunoglobulins, complement proteins, cytokines, growth factors, and oxidative stress in pathogenesis of early ARMD have been established in recent years [3]. The accumulation of extracellular plaques and deposits reveals a chronic inflammation that aggravates the effects of primary pathogenic stimuli [4].
Associations between ARMD and biochemical factors such as high sensitivity C-reactive protein (hs-CRP) have also been demonstrated [5]. In addition to hs-CRP, increased neutrophil level and lymphopenia have recently been investigated as biomarkers of inflammatory activity [6,7].
Inflammation is an important component of many age-related diseases such as Alzheimer disease and atherosclerosis [6]. The role of neutrophil-to-lymphocyte ratio (NLR) in cancer and cardiovascular disease and cancer has been reported [6,7], but its relationship with ocular diseases is unclear.
Given the role of inflammation in the pathogenesis of ARMD, this study aimed to investigate whether elevated NLR level could act as a risk factor of development of neovascular ARMD. Therefore, NLR and hs-CRP levels were evaluated as prognostic inflammatory biomarkers in ARMD patients.
The study was conducted by the ophthalmology department at Dr. Sami Ulus Maternity and Children's Health and Diseases Training and Research Hospital, after obtaining institutional review board and ethics committee approval. The study took place from September 2012 to January 2014. Informed consent was obtained from each subject prior to the study, and the described research adhered to the tenets of the Declaration of Helsinki. Forty patients with non-neovascular ARMD (group 1) and 40 patients with neovascular ARMD (group 2) were compared with 40 control participants without ARMD (group 3). Patients with hematologic disorders, acute or chronic infection, other inflammatory ocular and systemic disease, any ocular medication use, chronic obstructive pulmonary disease, current steroid therapy and/or history of steroid use 3 months prior to admission, or a history of cancer (with or without radiation or chemotherapy treatment) were excluded from the study. Visual acuity, biomicroscopic anterior and posterior segment examination, and intraocular pressure measurements were evaluated in all participants. All patients had been previously diagnosed with ARMD prior to admission to our hospital. No treatment such as intravitreal or laser treatment had yet been administered to any of the included patients, who presented to our ophthalmology clinic to verify their diagnoses of ARMD in other centers or to receive a prescription for eye drops.
Patients in group 2 demonstrated evidence of choroidal neovascular membranes or pigment epithelial detachments. Furthermore, a fluorescein angiogram and optical coherence tomography data at the time of initial diagnosis were required. Group 1 patients demonstrated evidence of drusen in the macula, with or without signs of geographic atrophy, in at least one eye. Patients with all other maculopathies were excluded from the study.
The NLR values were evaluated using an Horiba ABX Pentra DF 120 (Holliston, MA, USA) and hs-CRP levels were measured using Immage 800 (Beckman Coulter, Brea, CA, USA). The NLR levels of the patients were obtained from the hospital laboratory archive at the time of angiography and optical coherence tomography testing at the beginning of the study. The NLR was calculated by dividing the neutrophil count by the lymphocyte count.
All analyses were performed using PASW ver. 18.0 (SPSS, Chicago, IL, USA). Quantitative variables were expressed as mean ± standard deviation. Continuous variables were analyzed for normal distribution using the Kolmogorov-Smirnov test and analyzed for homogeneity using Levene's test. Comparisons of parametric values among groups were performed using one-way ANOVA. Comparisons of non-parametric values among groups were performed with the Kruskal-Wallis test. Tukey honest significant difference tests (for parametric variables) and Bonferroni adjustment after Mann-Whitney U-tests (for non-parametric variables) were used as post hoc tests for multiple comparisons between the groups. A two-tailed p-value less than 0.05 was considered significant. After performing univariate analysis, significant variables (age, gender, diabetes mellitus, NLR, and hs-CRP) were used in multivariate logistic regression analysis. Receiver operating characteristics curve analysis was performed to demonstrate the sensitivity and specificity of admission NLR and the optimal cutoff value for predicting neovascular ARMD.
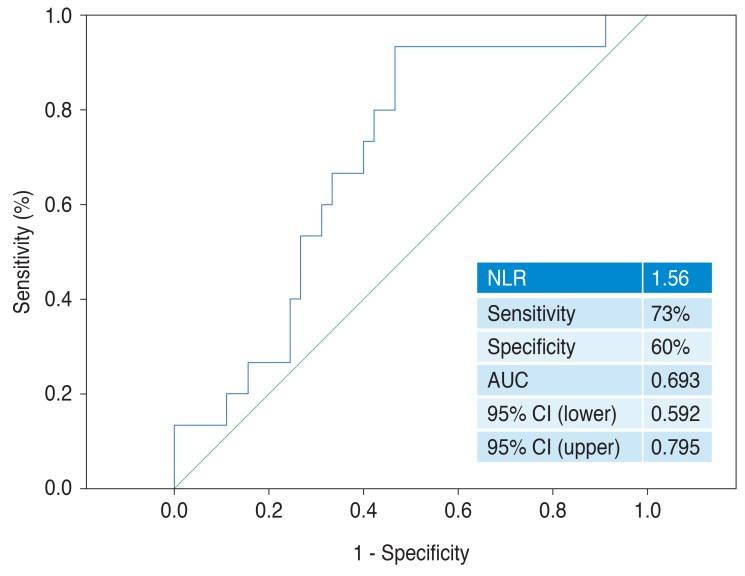
There were 20 female (50%) and 20 male (50%) patients both in group 1 and group 2 and 28 females (70%) and 12 males (30%) in group 3 (p = 0.051). The mean age was 65 ± 7 years in group 1, 75 ± 7 years in group 2, and 64 ± 6 years in group 3 (Table 1). The mean age was significantly higher in group 2 compared to groups 1 and 3 (p < 0.001). The ARMD was unilateral in 38 patients and bilateral in 42 patients. The NLR level was 1.65 ± 0.71 in group 1, 1.98 ± 0.84 in group 2, and 1.46 ± 0.44 in group 3. The hs-CRP value was 1.98 ± 0.251 mg/L in group 1, 3.242 ± 0.211 mg/L in group 2, and 1.145 ± 0.193 mg/L in group 3. Both NLR and hs-CRP values were significantly higher in group 2 compared to group 3 (p = 0.002 and p = 0.002, respectively) (Table 1). The number of people with diabetes mellitus was significantly higher in group 2 than groups 1 and 3 (p < 0.001). Baseline clinical features of the patients are shown in Table 1. In multivariate analysis, NLR remained an independent predictor of neovascular ARMD (odds ratio, 3.882; 95% confidence interval, 1.574 to 9.576; p = 0.003) together with age (p < 0.001), diabetes mellitus (p = 0.041), and hs-CRP (p = 0.018) (Table 2). The area under the receiver operating characteristics curve for NLR was 0.693, and an NLR of 1.56 or higher predicted neovascular ARMD with a sensitivity of 73% and specificity of 60% (Fig. 1).
The neovascularization period includes production and release of angiogenic factors, binding of factors to extracellular receptors, activation of intracellular signaling, endothelial cell activation, proliferation and migration, basement membrane degradation, remodeling of extracellular matrix, tube formation, and vascular stabilization [8]. In the treatment of ARMD, protection against oxidative stress and accumulation of lipofuscin and reduction and elimination of chronic inflammation are considered [8,9]. Oral and injectable anti-inflammatory medications are found to reduce the risk of ARMD through these mechanisms [10].
Many treatments such as laser photocoagulation, photodynamic therapy and intravitreal triamcinolone acetonide, and anti-vascular endothelial growth factor treatments to reduce vascular retina pigment epithelial permeability have been used to treat choroidal neovascularization secondary to ARMD. Triamcinolone acetonide takes part in the stabilization of the blood-retinal barrier and the downregulation of inflammation, in addition to demonstrating anti-angiogenic and anti-fibrotic properties [11]. Laser photocoagulation is no longer a standard of care treatment in the management of choroidal neovascularization [11]. In the treatment of ARMD, the potential use of celecoxib with its anti-inflammatory and anti-angiogenic effects has recently been emphasized [8,10,12].
All of these treatment modalities target the inflammation involved in the pathogenesis of ARMD. However, no clinical laboratory tests are used to establish the presence of such inflammation. Any test that reflects the inflammatory status of ARMD patients and its correlation with disease status would be a great innovation for monitoring treatment response in such patients.
NLR has been used for evaluation of inflammation in several cardiac and noncardiac diseases [13]. NLR calculation has been used to predict vascular calcification in end-stage renal disease patients [13]. Ahsen et al. [14] identified a relationship between Familial Mediterranean fever and NLR. As a result of such studies, NLR is now defined as an indicator of subclinical inflammation.
CRP is a non-specific marker of inflammation. Previous studies have demonstrated an association between elevated serum hs-CRP and homocysteine level in neovascular ARMD patients [5,15,16,17]. Cumulative oxidative damage caused by altered uric acid metabolism and hs-CRP could also play a role in the damage resulting from ARMD [1,18]. Our study shows the significant association between serum hs-CRP and neovascular ARMD.
A relationship between NLR and diabetic retinopathy has also been demonstrated [19]. A significant association between diabetes mellitus and neovascular ARMD was also identified in our study.
The assessment of NLR in patients with ARMD was also studied by Ilhan et al. [20]. In their study, ARMD patients were found to have higher NLR compared with controls. Furthermore, they found that NLR correlates with age and disease severity. In contrast to our study, they found significant difference between ARMD subgroups. This difference could be due to the small sample of patients of our study; additional studies are necessary to determine the associations of disease severity and duration of ARMD with NLR value. Ilhan et al. [20] also mentioned that NLR can be used as a biomarker of inflammation in ARMD. Older age and diabetes mellitus as risk factors of elevated NLR value in neovascular ARMD were demonstrated in our study.
Inflammation, vascular stress, and oxidative stress play important roles in the damage and pathogenesis of ARMD. As a result, elevated NLR level is a potential indicator of increased inflammatory activity in patients with ARMD. As a simple, inexpensive, and reliable prognostic biomarker, NLR level can be evaluated along with hs-CRP in ARMD patients. The NLR might also be used as a biomarker of response to ARMD treatments and for evaluation of prognosis. Further investigations including large populations are needed to investigate the possible role of NLR level in ARMD patients.
Notes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
REFERENCES
1. Subramani S, Khor SE, Livingstone BI, Kulkarni UV. Serum uric acid levels and its association with age-related macular degeneration (ARMD). Med J Malaysia 2010;65:36-40.

2. Coral K, Raman R, Rathi S, et al. Plasma homocysteine and total thiol content in patients with exudative age-related macular degeneration. Eye (Lond) 2006;20:203-207.


3. Rodrigues EB. Inflammation in dry age-related macular degeneration. Ophthalmologica 2007;221:143-152.


4. Anderson DH, Mullins RF, Hageman GS, Johnson LV. A role for local inflammation in the formation of drusen in the aging eye. Am J Ophthalmol 2002;134:411-431.


5. Boekhoorn SS, Vingerling JR, Witteman JC, et al. C-reactive protein level and risk of aging macula disorder: the Rotterdam Study. Arch Ophthalmol 2007;125:1396-1401.


6. Kaya MG. Inflammation and coronary artery disease: as a new biomarker neutrophil/lymphocyte ratio. Turk Kardiyol Dern Ars 2013;41:191-192.


7. Guthrie GJ, Charles KA, Roxburgh CS, et al. The systemic inflammation-based neutrophil-lymphocyte ratio: experience in patients with cancer. Crit Rev Oncol Hematol 2013;88:218-230.


8. Musat O, Ochinciuc U, Gutu T, et al. Pathophysiology and treatment of ARMD. Oftalmologia 2012;56:45-50.
9. Danulescu R, Costin D. The assessment of treatment efficacy in age related macular degeneration by evaluating the oxidative stress markers and OCT measurements. Rev Med Chir Soc Med Nat Iasi 2013;117:328-333.

10. Swanson MW, McGwin G Jr. Anti-inflammatory drug use and age-related macular degeneration. Optom Vis Sci 2008;85:947-950.


11. Becerra EM, Morescalchi F, Gandolfo F, et al. Clinical evidence of intravitreal triamcinolone acetonide in the management of age-related macular degeneration. Curr Drug Targets 2011;12:149-172.


12. Amrite AC, Kompella UB. Celecoxib inhibits proliferation of retinal pigment epithelial and choroid-retinal endothelial cells by a cyclooxygenase-2-independent mechanism. J Pharmacol Exp Ther 2008;324:749-758.


13. Turkmen K, Ozcicek F, Ozcicek A, et al. The relationship between neutrophil-to-lymphocyte ratio and vascular calcification in end-stage renal disease patients. Hemodial Int 2014;18:47-53.


14. Ahsen A, Ulu MS, Yuksel S, et al. As a new inflammatory marker for familial Mediterranean fever: neutrophil-to-lymphocyte ratio. Inflammation 2013;36:1357-1362.


15. Seddon JM, Gensler G, Milton RC, et al. Association between C-reactive protein and age-related macular degeneration. JAMA 2004;291:704-710.


16. Seddon JM, Gensler G, Klein ML, Milton RC. C-reactive protein and homocysteine are associated with dietary and behavioral risk factors for age-related macular degeneration. Nutrition 2006;22:441-443.


17. Nowak M, Swietochowska E, Wielkoszynski T, et al. Homocysteine, vitamin B12, and folic acid in age-related macular degeneration. Eur J Ophthalmol 2005;15:764-767.

18. Mehryar M, Farvardin M, Hosseini H, Aslani M. Potential role of uric acid in the molecular pathogenesis of age-related macular degeneration. Med Hypotheses 2006;66:793-795.


Fig. 1
The receiver operating characteristics analysis for neutrophil-to-lymphocyte ratio (NLR) in predicting neovascular age-related macular degeneration. AUC = area under the curve; CI = confidence interval.
- TOOLS
-
METRICS

- Related articles
-
The Relationship between Myopia and Obesity in Adults2024 April;38(2)





 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



