Dry eye disease is a multifactorial disorder of the tears and ocular surface [1]. It leads to various ocular symptoms and tear film instability induce potential damage of the ocular surface and ocular surface inflammation [1]. The resulting diminished vision and quality of life affect millions of people worldwide [2]. Treatments for dry eye disease in increasing order of severity include artificial tears, anti-inflammatory agents, immunosuppressants, punctal plugs, serum eye drops, contact lenses, and surgery [3,4]. Artificial tears are typically used as the first-line management of dry eye disease. They improve the stability of the tear film and provide symptomatic relief [5].
Hyaluronic acid (HA) is a linear polymer composed of long chains of repeating disaccharide units of N-acetylglucosamine and glucuronic acid [6]. HA is an avid moisturizer and has a higher residency time than other artificial tear components. In addition, HA can protect the ocular surface epithelium by facilitate epithelial healing [7]. 0.1% HA eye drops are commonly used to treat dry eye disease and were found to be effective in improving symptoms as well as signs including corneal epithelial injury [8,9,10]. However, due to a short duration of action and deficient tear lipid or mucin components, HA eye drops have several limitations including the need for frequent application.
Mineral oil (MO) is a complex mixture of saturated hydrocarbons derived from petroleum through various refining steps and subsequent purification [11]. MO-based artificial tears as an ointment formulation prolongs tear retention time in the eye, therefore requiring fewer daily applications [12]. However, no study has been performed on eye drop mixtures of HA and MO as an oil component for dry eye.
The purpose of this study was to evaluate the efficacy of MO and HA combination eye drops for the management of dry eye disease using a desiccating stress-induced mouse model, by evaluating the changes of tear production, tear film break-up time (TBUT), fluorescein staining, ocular surface irregularities, and goblet cell count in the conjunctiva.
Materials and Methods
Mouse model of dry eye and experimental procedure
The research protocol was approved by the Chonnam National University Medical School Research Institutional Animal Care and Use Committee. All animals were treated according to the standards in the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research. Six- to eight-week-old female C57BL/6 mice were used in these experiments.
Experimental dry eye (EDE) was induced by subcutaneous injection of 0.5 mg/0.2 mL scopolamine hydrobromide (Sigma-Aldrich, St. Louis, MO, USA) four times a day (9 a.m., 1 p.m., 5 p.m., and 9 p.m.) with exposure to an air draft and 30% ambient humidity [13,14,15,16]. During these experiments, the animal's behavior, food, and water intake were not restricted.
The mice were randomly divided into six groups (n = 5 per group) depending on topical treatment administered: untreated (UT) mice; EDE mice that received no eye drops; EDE mice treated with non-preservative 0.1% HA eye drops (Alcon Korea, Seoul, Korea); EDE mice treated with 0.1% MO and 0.1% HA mixture eye drops; EDE mice treated with 0.5% MO and 0.1% HA mixture eye drops; and EDE mice treated with 5.0% MO and 0.1% HA mixture eye drops. MO and HA mixture eye drops were created by mixing MO with 0.1% HA using a surfactant. All treatment groups received 2 µL of eye drops four times a day.
At 5 and 10 days after treatment, tear volume, corneal irregularity score, TBUT, and corneal fluorescein staining score were measured two hours after application of the last eye drops. Ten days after treatment, the mice were euthanized, and Periodic acid-Schiff staining was performed. Each experiment was repeated three times.
Measurement of tear volume
Tear volume was assessed using phenol-red impregnated Zone-Quick cotton threads (Oasis, Glendora, CA, USA) as previously described [17,18,19]. Cotton threads were placed in the lateral canthus for 20 seconds. The threads length that became wet by tears was measured using a SMZ 1500 stereoscopic zoom microscope (Nikon, Melville, New York, NY, USA). A standard curve was derived to convert distance into volume.
Evaluation of corneal surface irregularity score
The severity of the corneal surface irregularity was graded by measuring the distortion of a white ring from the fiber-optic ring illuminator of the stereoscopic zoom microscope by two blinded observers [18,19]. The corneal irregularity score was calculated using a 6-point scale (0-5) based on the number of distorted quarters in the reflected ring, as follows: 0, no distortion; 1, distortion in one quarter of the ring; 2, distortion in two quarters; 3, distortion in three quarters; 4, distortion in all four quadrants; and 5, severe distortion, in which no ring could be recognized [20].
Measurement of tear film break-up time
One micro-liter of 0.1% liquid sodium fluorescein was gently applied to the conjunctival sac. After 3 blinks, the interval between the last complete blink and the appearance of the first corneal black spot was recorded in seconds using a slit-lamp microscope equipped with a cobalt blue filter. And the mean of three measurements was calculated [21,22].
Evaluation of the fluorescein staining score
The severity of corneal epithelial damage was graded by two blinded observers who measured the fluorescein staining of the mouse cornea. After 0.1% liquid sodium fluorescein was dropped into the conjunctival sac, the corneal epithelial damage was graded with a slit-lamp microscope equipped with a cobalt blue filter. The fluorescein staining score was calculated using a 5-point scale (0-4), as follows: 0, no fluorescein staining; 1, slightly punctuate staining <30 spots; 2, punctuate staining >30 spots, but not diffuse; 3, severe diffuse staining but no positive plaque; and 4, severe diffuse staining with positive fluorescein plaque [22]. After scoring all four corneal quadrants, the total score was averaged.
Histology
Eyes and adnexa were surgically excised, fixed in 4% paraform-aldehyde, and embedded in paraffin. Six-micrometer sections were stained with Periodic acid-Schiff reagent. Sections from each group were examined and photographed with a microscope (BX53; Olympus, Tokyo, Japan) equipped with a digital camera (F2; Foculus, Finning, Germany). Goblet cells in the conjunctiva were counted in three sections from each eye using image analysis software (Media Cybernetics, Silver Spring, MD, USA) and expressed as the number of goblet cells per 100 µm [18,19].
Statistical analysis
Statistical differences in the tear volume, corneal irregularity score, TBUT, and fluorescein staining score were evaluated by one-way analysis of variance, with post hoc analysis. Kruskal-Wallis and Mann-Whitney tests were used to compare the levels of cytokines and chemokine between different groups. A p-value <0.05 was considered statistically significant.
Results
Aqueous tear production
The mean tear volumes at 5 days after desiccating stress were 0.035 ± 0.004 µL in the UT group, 0.013 ± 0.003 µL in the EDE group, 0.014 ± 0.002 µL in the HA group, 0.013 ± 0.002 µL in the mixed 0.1% MO group, 0.017 ± 0.003 µL in the mixed 0.5% MO group, and 0.013 ± 0.002 µL in the mixed 5.0% MO group. There were no significant differences in the tear volumes between groups. At 10 days, the mean volume in all groups showed similar finding to those at 5 days.
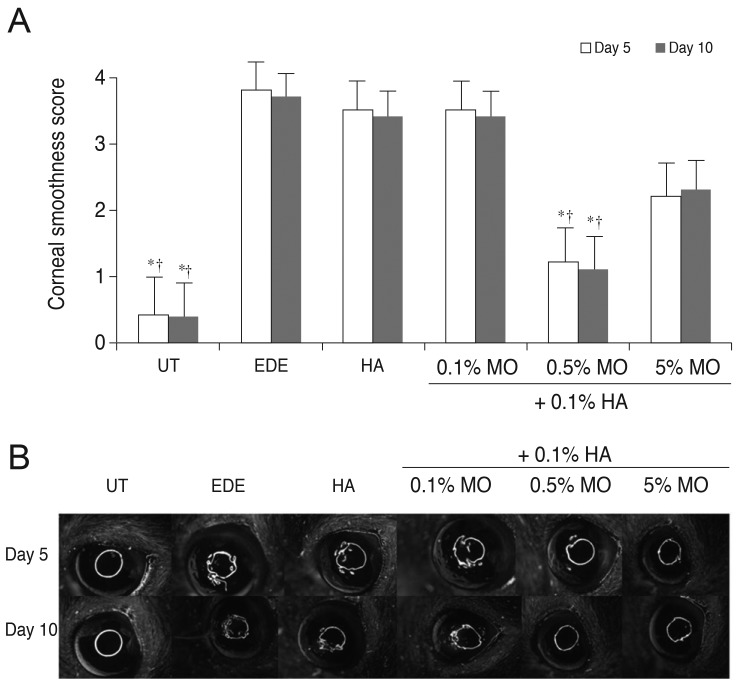
Corneal surface irregularity score
Corneal irregularity scores increased from 0.25 ± 0.45 to 3.92 ± 0.90 (p < 0.01) at 5 days after desiccating stress. The mean corneal irregularity scores at 5 days after treatment were 3.46 ± 0.52 in the HA group (p = 0.51 compared with the EDE group), 3.36 ± 0.51 in the mixed 0.1% MO group (p = 0.30 vs. the EDE group; p = 0.99 vs. the HA group), 1.27 ± 0.47 in the mixed 0.5% MO group (p < 0.01 vs. the EDE and HA groups), and 3.27 ± 0.79 in the mixed 5.0% MO group ( p = 0.16 vs. the EDE group; p = 0.98 vs. the HA group). The results for corneal irregularity scores in all groups at 10 days after desiccating stress were similar to those at 5 days (Fig. 1).
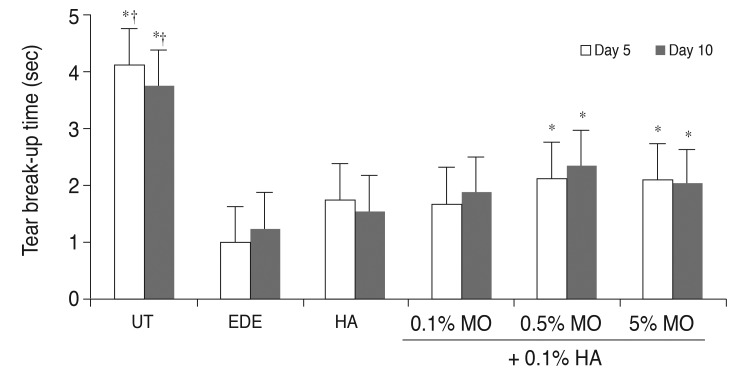
Tear film break-up time
In the UT group, TBUT was 4.13 ± 0.58 and 3.75 ± 1.04 seconds at 5 and 10 days respectively. After desiccating stress, the mean TBUT in the EDE group was 1.00 ± 1.04 and 1.26 ± 0.54 seconds at 5 and 10 days, respectively (p < 0.05 compared with the UT group for both). In the treatment group, the TBUT values at 5 days were 1.76 ± 1.04 seconds in the HA group (p = 0.31 vs. the EDE group), 1.67 ± 1.16 seconds in the mixed 0.1% MO group (p = 0.40 vs. the EDE group; p = 0.10 vs. the HA group), 2.13 ± 0.60 seconds in the mixed 0.5% MO group (p = 0.02 vs. the EDE group; p = 0.93 vs. the HA group), and 2.10 ± 0.76 seconds in the mixed 5.0% MO group (p = 0.01 vs. the EDE group; p = 0.93 vs. the HA group). At 10 days, TBUT in all groups showed similar findings to those at 5 days (Fig. 2).
Corneal fluorescein staining score
After desiccating stress, corneal fluorescein staining scores at 5 and 10 days were 3.00 ± 0.93 and 2.86 ± 0.64 in the EDE group (p < 0.01 compared with the UT group for both). After treatment, the mean staining scores were 2.10 ± 0.32 in the HA group (p < 0.01 vs. the EDE group), 1.27 ± 0.47 in the mixed 0.1% MO group (p < 0.01 vs. the EDE group; p = 0.03 vs. the HA group), 1.18 ± 0.60 in the mixed 0.5% MO group (p < 0.01 vs. the EDE group; p = 0.01 vs. the HA group), 3.33 ± 0.49 seconds in the mixed 5.0% MO group ( p = 0.67 vs. the EDE group; p < 0.01 vs. the HA group). The results for corneal fluorescein staining scores in all groups at 10 days after desiccating stress were similar to those at 5 days (Fig. 3).
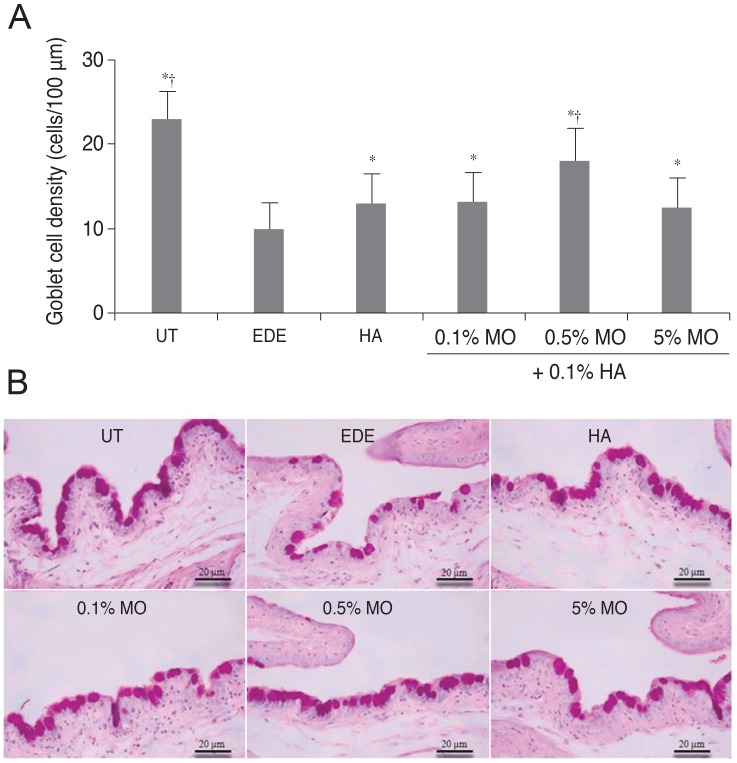
Conjunctival goblet cell count
The mean conjunctival goblet cell count significantly decreased in the EDE group (10.0 ± 0.8 cells/100 µm) compared with the UT group (23.0 ± 1.4 cells/100 µm) (p < 0.01). The mean goblet cell counts were 13.0 ± 1.41 cells/100µm in the HA group (p = 0.02 vs. the EDE group), 13.3 ± 0.96 cells/100 µm in the mixed 0.1% MO group (p < 0.01 vs. the EDE group; p = 0.78 vs. the HA group), 18.0 ± 0.82 cells/100 µm in the mixed 0.5% MO group (p < 0.01 vs. the EDE group; p = 0.02 vs. the HA group), and 12.5 ± 1.29 cells/100 µm in the mixed 5.0% MO group (p = 0.02 vs. the EDE group; p = 0.62 vs. the HA group) (Fig. 4).
Discussion
HA can be found naturally in all vertebrates in the extracellular matrix of the skin, synovial fluid, and vitreous body of the eye. It is a biopolymer of disaccharide units composed of N-acetylglucosamine and glucuronic acid in linear chains of varying molecular weights [23]. By forming a protective coating that help prevent further irritation and d amage of the corneal epithelium, HA can improve corneal fluorescein staining [24]. In addition, dose-dependent ability of HA to decrease the size of the wound area has been reported [25]. Topical use of HA provides both objective and subjective symptomatic relief in patients with dry eye disease [8,9,10]. In addition, there is evidence that hyaluronate may play a role in controlling the localized inflammation of ocular surface in patients with keratoconjuctivitis sicca [26].
HA has several biological effects that are exerted through its direct interaction with a cell surface receptor (CD44). The effects include metastatic potential of tumor cells, secretion of cytokines and chemokines, mediation of inflammatory responses, and cell division [27,28,29]. In addition, binding of HA to CD44 enhances the growth of the corneal epithelial cells and promotes migration of human corneal epithelial cells [27,28]. The expression of CD44, the receptor of HA, can be increased in corneal and conjunctival cells of patients with dry eye disease, whereas it is decreased following the use of HA eye drops [24,30].
Different types of artificial tear formulation, including gel-based, cellulose-based, carbomer-based and MO-based, have been developed to relieve symptoms of dry eye and have been designed as alternatives to classic artificial tear formulation [12,31,32]. Among these new formulations, MO-based formulations can prolong retention times compared with aqueous tear substitutes, but they may cause a sticky sensation and blurred vision [33]. One study demonstrated that a preservative-free oil-in-water emulsion containing 7% soy bean oil, 3% egg yolk phospholipids, and1.8% glycerol could relieve and improve clinical signs such as tear volume and corneal fluorescent staining in a mouse model of dry eye [34]. In addition, MO-based artificial tears were as effective as cellulose-based and carbomer-based artificial tears in reducing subjective symptoms, and objective signs including TBUT [12].
In the present study, we evaluated the effect of mixed 0.1% HA and MO eye drops on tear production, ocular surface irregularity, TBUT, corneal fluorescein staining score, and conjunctival goblet cell count in an EDE model. There was no significant difference of tear volume between the stress-induced groups. The corneal irregularity score was lower in the mixed 0.5% MO group compared with the EDE and HA groups. The mixed 0.5% and 5.0% MO groups showed a significant improvement in TBUT compared with the EDE group. In the mixed 0.1% and 0.5% MO groups, there was a significant improvement of the corneal fluorescein staining score compared with the EDE or HA groups. The conjunctival goblet cell count was higher in the mixed 0.5% MO group compared with the EDE group and the HA group.
Our results showed that HA eye drops containing MO could have a beneficial effect on the tear film and ocular surface of EDE. These results may be attributed to a synergetic effect of HA and MO. The effect of HA as artificial tears which could decrease corneal fluorescein staining by promoting epithelial healing might be enhanced by the addition of MO, and MO itself as a lipid component might improve tear stability and corneal surface irregularity by prolonging the retention time of the tear film. The 0.1% MO showed an improvement of corneal fluorescein staining scores and goblet cell count, and the 5.0% MO improved the TBUT and goblet cell count, while the 0.5% MO ameliorated all measured parameters. These results suggested that the application of a mixture of 0.5% MO and HA eye drops was the best combination for the treatment of EDE compared with the other MO groups.
The application of MO eye drops, however, may cause a sticky, burning sensation and blurred vision [33]. Because this study is based on the dry eye mouse model, further studies based on human will be needed to examine any possible side effects.
In conclusion, the application of eye drops containing a mixture of 0.5% MO and 0.1% HA was more effective in corneal irregularity score, corneal fluorescein staining score, and conjunctival goblet cell count on the ocular surface compared with 0.1% HA eye drops in a mouse model of dry eye. These results showed that the MO and HA mixture was more effective than HA agents alone for treating dry eye associated with ocular surface damage. In the near future, clinical studies on the safety and efficacy of MO and HA mixture eye drops for the treatment of dry eye will be needed.




 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print






