 |
 |
| Korean J Ophthalmol > Volume 28(3); 2014 > Article |
Abstract
Purpose
To evaluate the usefulness of the interferon-gamma release assay (IGRA) for diagnosing tuberculosis (TB)-related uveitis (TRU).
Methods
Records from 181 patients with ocular signs and symptoms suggestive of TRU and intraocular inflammation of unknown etiology were reviewed. All subjects underwent clinical and laboratory testing, including IGRA, to rule out presence of underlying disease. A diagnosis of presumed TRU was made based on an internist's TB diagnosis and a patient's response to anti-TB therapy. Sensitivity, specificity, and positive predictive values of IGRA for TRU diagnosis were calculated. Clinical characteristics were compared between patients with positive and negative IGRA results.
Results
The sensitivity and specificity of IGRA for TRU were 100% and 72.0%, respectively. Mean age, percentage of patients with retinal vasculitis, and the anatomic type of uveitis were significantly different between patients with positive and negative IGRA results (all p Ōēż 0.001). Positive IGRA rates and false-positive rates were significantly different between age and anatomic type groups (both p = 0.001). The positive predictive value of the IGRA among patients with intraocular inflammation was high (70%) when all of younger age (Ōēż40 years), posterior uveitis, and retinal vasculitis were present.
Conclusions
The IGRA is useful for diagnosing TRU in the Korean population, especially when it is used as a screening test. Clinical characteristics, including younger age (Ōēż40 years), posterior uveitis, and retinal vasculitis in IGRA-positive patients, increase the likelihood of the patient having TRU.
Tuberculosis (TB) is the single most common cause of morbidity and mortality worldwide, but it is also the most cost-effective disease to treat [1,2,3]. TB is caused by Mycobacterium tuberculosis (MTB), which causes chronic and indolent systemic illness in multiple organs of the body [4]. Most patients with TB remain symptom-free, and illness occurs in only 3% to 5% of infected people. The incidence of TB-related uveitis (TRU) in patients with intraocular inflammation varies between 0.6% and 10.5%, depending on the country [4].
Cases of TRU are generally chronic and progress slowly. As MTB requires oxygen to grow, it tends to remain in the choroid and ciliary body. However, it can be present in any part of the eye and thus, the condition presents in variable ways. TRU most commonly manifests as anterior uveitis, multifocal choroiditis, panuveitis, or vasculitis. The most accurate diagnostic tool to confirm TRU is the acid-fast smear/culture combination, followed by polymerase chain reaction of an ocular fluid sample. Unfortunately, obtaining an ocular fluid sample of sufficient volume is difficult in most cases and these tests are not highly specific. Although the current gold standard for TB diagnosis is a positive tuberculin skin test (TST) combined with appropriate clinical findings, thi s test has somewhat limited accuracy. The TST can have a high false-positive rate in Korea, which has an intermediate TB burden and a large number of Bacille Calmette-Gu├®rin (BCG)-vaccinated individuals [2,5].
Among other diagnostic tests, the interferon-gamma release assay (IGRA) has been reported to be useful for detecting TB infections [5,6]. Kang et al. [5] showed that 95.7% of Korean subjects had received the BCG vaccine, and that 51% and 4% of subjects had a positive TST and IGRA, respectively, even though the study group had no identifiable risk factors for TB. This result indicates that IGRA is a better TB test than the TST in Korea. Interestingly, Ang et al. [7] found that IGRA was more specific than TST, and the high TST false-positive rate in BCG-vaccinated subjects favors the use of IGRA. Itty et al. [6] also suggested that IGRA is useful in detecting TRU, especially in immunocompromised or BCG-vaccinated patients.
However, the IGRA has a high cost, technical limitations, and a limited ability to differentiate between active and latent TB. Although several studies have shown the usefulness of IGRA in detecting TRU, few studies have investigated which patients should be tested with IGRA. Clinical factors may affect IGRA results, including age (Ōēź46 years) and TB history, both of which are independent risk factors for false-positive outcomes [8]. However, a recent study by Critselis et al. [9] showed that a positive IGRA result was not associated with age in children investigated for latent TB infections.
The aim of the present study was to evaluate the usefulness of IGRA for diagnosing TRU in Korean patients with intraocular inflammation, and to assess the effect of clinical factors, including age, on IGRA results. Clinical factors were compared between patients with and without positive IGRA results as well as between patients with and without presumed TRU. Furthermore, positive IGRA rates were compared among subgroups created based on specific clinical factors. Lastly, associations between clinical factors and IGRA results were assessed using multiple regression analyses.
All aspects of this research protocol adhered to the tenets of the Declaration of Helsinki. The institutional review board of Seoul National University Bundang Hospital approved this study and waived the need for informed consent. Included patients had clinical findings consistent with TRU [4] and intraocular inflammation of unknown infectious cause. The medical records of 246 consecutive patients with intraocular inflammation who visited Seoul National University Bundang Hospital between February 2010 and June 2012 were retrospectively reviewed. Among these patients, 238 had various types of intraocular inflammation and were screened with the IGRA. Additional laboratory tests were performed to identify possible causes of uveitis and included a complete blood count, serum chemistry analysis, serology, urine analysis, chest and pelvis radiography, rheumatoid factor evaluations, antinuclear antibody, and human leukocyte antigen-B27 levels. Fifty-three patients were excluded because of confirmed non-TB causes of intraocular inflammation, including toxoplasmosis, toxocariasis, and herpetic keratouveitis. An additional four patients were excluded because of indeterminate IGRA results. Therefore, a total of 181 patients were included in the study analyses. Slit lamp examination, fundus examination, optical coherence tomography, and fluorescein angiography were performed and all results were evaluated by two experienced retinal specialists (SJW and KHP).
Patients were asked about recent fevers, night sweats, pulmonary or extrapulmonary symptoms, and weight loss. Additionally, any history of TB or immunosuppression was discussed and a thorough review of respiratory, gastrointestinal, neurological, genitourinary, and musculoskeletal systems was performed. Patients with suspected TB uveitis were referred to internists at Seoul National University Bundang Hospital, who made independent TB diagnoses using standard recommended guidelines [10]. Following identification of a systemic TB infection and exclusion of other uveitis causes, a diagnosis of presumed TRU (Fig. 1) was made. Patients with presumed TRU were treated with the standard daily four drug antimicrobial therapy cocktail (5 mg/kg isoniazid, 10 mg/kg rifampicin, 15 mg/kg ethambutol, and 25 mg/kg pyrazinamide) for 2 to 3 months followed by 4 to 10 months of daily 5 mg/kg isoniazid and 10-mg/kg rifampicin. In addition, 0.5-mg/kg oral prednisolone was administered daily for the first week of therapy and then tapered over 3 weeks. To support the TRU diagnosis, patients were examined 4 weeks after initiating anti-TB therapy to confirm improvements in ocular inflammation and/or fundoscopic signs. Patients were not classified as having presumed TRU if remission was not achieved with antimicrobial therapy after 6 months.
The QuantiFERON-TB Gold blood test (Cellestis Limited, Carnegie, VIC, Australia) was used for IGRA testing and is an in vitro laboratory diagnostic test used on whole blood specimens. It contains peptide cocktails that simulate ESAT-6 and CFP-10 proteins, which are absent from all BCG strains and from most non-TB mycobacteria. The test relies on the principle that previously sensitized T-cells produce interferon-gamma upon re-exposure [11,12,13].
Patients screened with the IGRA were divided into the following four groups, according to the primary location of ocular inflammation: anterior uveitis, intermediate uveitis, panuveitis, and posterior uveitis. Clinical features, including age, sex, anatomic type, and TB history, were compared between patients with positive and negative IGRA results using the Fisher's exact or the Student's t-test. Positive IGRA rates were calculated among age (group 1, 0 to 30 years; group 2, 31 to 40 years; group 3, 41 to 50 years; group 4, 51 to 60 years; group 5, 61 to 80 years) and anatomic type subgroups. Clinical features were compared between presumed TRU and non-TRU patients.
Continuous variables are presented as mean ┬▒ standard deviation throughout the manuscript. Statistical analyses were performed using the SPSS ver. 17.0 (SPSS Inc., Chicago, IL, USA). Student's t-tests and analyses of variance were used for continuous variables, while the chi-square test was used for dichotomous variables. Statistical significance was defined as a p-value <0.05.
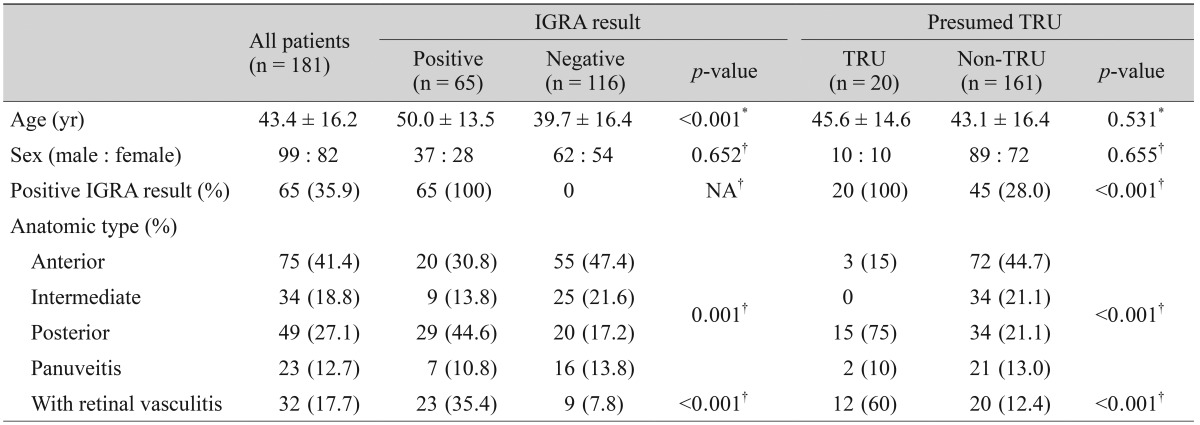
Clinical characteristics of the 181 patients (99 men and 82 women) included in the analyses are presented in Table 1. Mean patient age was 43.4 ┬▒ 16.2 years (range, 6 to 80 years). Among the 181 patients with intraocular inflammation, 65 (35.9%) had a positive IGRA result. Additionally, 20 patients (11.0%) were diagnosed with presumed TRU because high resolution computed tomography (HRCT) results or a positive acid-fast bacteria smear/MTB culture were suggestive of a pulmonary TB infection. All patients with presumed TRU had a positive IGRA test (IGRA sensitivity for presumed TRU = 100%) and 116 of 161 patients without presumed TRU had negative IGRA results (IGRA specificity for presumed TRU = 72.0%).
Patient age was significantly different between patients with positive and negative IGRA results (50.0 ┬▒ 13.5 years in positive patients, 39.7 ┬▒ 16.4 years in negative patients, p < 0.001). Patients with positive and negative IGRA results also had significantly different anatomic types of inflammation. Remarkably, posterior uveitis was noted in 44.6% of patients with a positive IGRA result, but in only 17.2% of patients with a negative result. Furthermore, the proportion of patients with retinal vasculitis was significantly higher among patients with a positive IGRA result (35.4%) than among those with a negative IGRA result (7.8%, p < 0.001). Patient age was not significantly different between patients with (45.6 ┬▒ 14.6 years) and without (43.1 ┬▒ 16.4 years) presumed TRU (p = 0.531). However, posterior uveitis and retinal vasculitis were more frequently noted in patients with presumed TRU (75% and 21.1%, respectively) than in those with intraocular inflammation from other causes (60% and 12.4%, respectively, p < 0.001).
Data were examined and compared in each of the five age subgroups and the percentage of IGRA-positive results in each group is presented in Fig. 2. The percentage of patients with positive IGRA results were 18.9%, 24.4%, 29.7%, 52.8%, and 60% in age groups 1, 2, 3, 4, and 5, respectively. The percentage of false-positive results were 11.8%, 13.9%, 27.8%, 39.3%, and 55.6%, in age groups 1, 2, 3, 4, and 5, respectively. Both true- and false-positive IGRA results were significantly different among age groups and were linearly correlated with age (i.e., true and false positive rate increased with age, p < 0.001, chi-square test for trend).
Positive IGRA result data were also analyzed among the four different inflammation location groups. Patients in the anterior uveitis, intermediate uveitis, posterior uveitis, and panuveitis groups had IGRA positive rates of 26.7%, 26.5%, 59.2%, and 30.4%, respectively (Fig. 3). More patients with retinal vasculitis (71.9%) had positive IGRA test results than patients without retinal vasculitis (28.2%, p < 0.001). False positives occurred in 15.0%, 0.0%, 51.7%, and 28.6% of patients with anterior, intermediate, posterior, and panuveitis, respectively. Positive IGRA results and false-positive rates were also statistically different among patients with different anatomic inflammation types (p = 0.001 and 0.002, respectively).
The positive predictive values for presumed TRU among patients with intraocular inflammation are shown in Fig. 4. Values were 42.9%, 50.0%, 9.1%, 42.1%, and 16.7% in age groups 1, 2, 3, 4, and 5, respectively. Values were 15.0%, 0.0%, 51.7%, and 28.6% in patients with anterior, intermediate, posterior, and panuveitis, respectively. In patients with and without retinal vasculitis, the positive predictive value of the IGRA was 52.2% and 22.8%, respectively. The IGRA test had greater positive predictive values in patients with a younger age (Ōēż40 years), posterior uveitis, and retinal vasculitis. The positive predictive value of the IGRA among patients with intraocular inflammation was 70% when all of these factors were present.
Multiple logistic regressions were performed to identify factors associated with the IGRA results. Age was independently associated with a positive IGRA result (p < 0.001), but the anatomic type of inflammation was not (p = 0.176). The odds ratio for age adjusted for presumed TRU and anatomic type was 1.06 (95% confidence interval [CI], 1.03 to 1.09), and that for age group adjusted for the same confounding variables was 1.90 (95% CI, 1.41 to 2.56; p < 0.001). Likelihood ratios were calculated for assessing the value of performing the IGRA diagnostic test. The positive likelihood ratio was 3.57 and the negative likelihood ratio was 0.00 in our study.
Intraocular inflammation can have many different origins and presents with a wide spectrum of clinical manifestations. If the underlying inflammatory cause is treatable, prompt and appropriate treatment can result in a favorable outcome. In the current study, we examined the IGRA as a diagnostic tool for presumed TRU in Korean patients with intraocular inflammation. Our results suggest that the IGRA is highly sensitive and moderately specific for TRU. A test used for TB screening should have good sensitivity and acceptable specificity [14], so we believe that IGRA can be used as a screening test for presumed TRU in Korea. Unfortunately, the TST (Mantoux test) is not useful as a screening tool because of the high false-positive rate in the Korean population, which has a very high BCG vaccination rate. Therefore, an alternative test is needed for the Korean population, and in this study, we evaluated the IGRA as a screening method for TRU. However, the false-positive rate (1-specificity) of IGRA was not low and cannot be neglected. Therefore, clinicians should consider which patients are more likely to have true TRU. Our analyses showed that younger age (Ōēż 40 years), a positive IGRA, and the presence of posterior uveitis and retinal vasculitis were all predictive of TRU.
Broad-based posterior synechiae, retinal vasculitis (with or without choroiditis), and serpiginous choroiditis in patients with latent or manifest TB are clinically suggestive of TRU in TB-endemic areas [15,16,17]. This may explain why the positive predictive value of IGRA was greater in patients with posterior uveitis and retinal vasculitis. Several studies [15,16,18,19] have reported that vasculitis can occur from a TB infection, and it may be associated with hypersensitivity to MTB [20].
We examined how a positive IGRA result should be interpreted in patients with suspected TB uveitis. Because our older patients tended to have a higher IGRA positivity rate, a positive result is likely insufficient to make a definitive TRU diagnosis, especially in elderly patients with intraocular inflammation. This result is comparable to those of Kang et al. [5], who reported that positive IGRA rates are positively and linearly correlated with patient age. In our study, the age of patients with and without TRU were not significantly different, so this relationship did not result from inherent group differences in age.
We also examined which patients should have the IGRA performed for diagnosing presumed TRU. Patients with intraocular inflammation usually undergo extensive diagnostic work-ups to identify the underlying inflammatory cause. Performing the IGRA on all patients with intraocular inflammation would not be cost-effective. Therefore, we recommend having the IGRA done for patients with a clinical presentation suggestive of TRU, including retinal vasculitis and posterior uveitis. In addition, because a positive IGRA result in a young patient with posterior uveitis or retinal vasculitis likely indicates TRU, the test should be heavily considered if a concurrent TB infection is suspected.
Several limitations of our study require consideration. Our calculations for positive and negative predictive values are limited by the absence of normal control data. Ang et al. [21] recommend that TRU be diagnosed by considering clinical presentation and the IGRA and TST results. Our internists did not perform the TST because prior BCG vaccination is associated with high false positive rates and the Korean population has widely received the BCG vaccine (95.6% of children [22], 95.7% of adults [5]). Therefore, we could not compare or relate TST and IGRA results in this study, even though this comparison is crucial for evaluating the relative diagnostic value of the IGRA. Additionally, further investigations are needed to evaluate the true relationship between positive IGRA result rates and age among normal, healthy control subjects. Lastly, prospective studies that include a larger number of patients may provide more concrete information on the usefulness of the IGRA in diagnosing TRU.
In conclusion, the IGRA may be a useful screening test for presumed TRU in Korea. Patients with positive results should be further evaluated with other reliable tests for TB (e.g., sputum culture, HRCT, anti-TB medication response) to rule-out or confirm TRU. Patient age and uveitis anatomic location may be helpful in interpreting the IGRA results with respect to a TRU diagnosis. In particular, a positive IGRA result is highly suggestive of TRU in Korea in a younger patient (Ōēż 40 years) with posterior uveitis and retinal vasculitis.
Acknowledgements
This work was supported by a grant (2012R1A2A2A02012821) funded by the National Research Foundation in Korea.
REFERENCES
1. Centers for Disease Control and Prevention (CDC). Trends in tuberculosis: United States, 2004. MMWR Morb Mortal Wkly Rep 2005;54:245-249.

2. Dye C, Scheele S, Dolin P, et al. WHO Global Surveillance and Monitoring Project. Consensus statement. Global burden of tuberculosis: estimated incidence, prevalence, and mortality by country. JAMA 1999;282:677-686.


3. Centers for Disease Control and Prevention (CDC). Expanded tuberculosis surveillance and tuberculosis morbidity: United States, 1993. MMWR Morb Mortal Wkly Rep 1994;43:361-366.

5. Kang YA, Lee HW, Yoon HI, et al. Discrepancy between the tuberculin skin test and the whole-blood interferon gamma assay for the diagnosis of latent tuberculosis infection in an intermediate tuberculosis-burden country. JAMA 2005;293:2756-2761.


6. Itty S, Bakri SJ, Pulido JS, et al. Initial results of QuantiFERON-TB Gold testing in patients with uveitis. Eye (Lond) 2009;23:904-909.


7. Ang M, Htoon HM, Chee SP. Diagnosis of tuberculous uveitis: clinical application of an interferon-gamma release assay. Ophthalmology 2009;116:1391-1396.


8. Feng Y, Diao N, Shao L, et al. Interferon-gamma release assay performance in pulmonary and extrapulmonary tuberculosis. PLoS One 2012;7:e32652



9. Critselis E, Amanatidou V, Syridou G, et al. The effect of age on whole blood interferon-gamma release assay response among children investigated for latent tuberculosis infection. J Pediatr 2012;161:632-638.


10. Diagnostic Standards and Classification of Tuberculosis in Adults and Children. This official statement of the American Thoracic Society and the Centers for Disease Control and Prevention was adopted by the ATS Board of Directors, July 1999. This statement was endorsed by the Council of the Infectious Disease Society of America, September 1999. Am J Respir Crit Care Med 2000;161(4 Pt 1):1376-1395.


11. Mazurek GH, Jereb J, Lobue P, et al. Guidelines for using the QuantiFERON-TB Gold test for detecting Mycobacterium tuberculosis infection, United States. MMWR Recomm Rep 2005;54(RR-15):49-55.

12. Mazurek GH, Jereb J, Vernon A, et al. Updated guidelines for using interferon gamma release assays to detect Mycobacterium tuberculosis infection: United States, 2010. MMWR Recomm Rep 2010;59(RR-5):1-25.
13. Madariaga MG, Jalali Z, Swindells S. Clinical utility of interferon gamma assay in the diagnosis of tuberculosis. J Am Board Fam Med 2007;20:540-547.


15. Gupta A, Bansal R, Gupta V, et al. Ocular signs predictive of tubercular uveitis. Am J Ophthalmol 2010;149:562-570.


16. Gupta A, Gupta V, Arora S, et al. PCR-positive tubercular retinal vasculitis: clinical characteristics and management. Retina 2001;21:435-444.


19. Reny JL, Challe G, Geisert P, et al. Tuberculosis-related retinal vasculitis in an immunocompetent patient. Clin Infect Dis 1996;22:873-874.


Fig.┬Ā1
Diagnosis of presumed tuberculosis (TB)-related uveitis. (A) Fundus photograph (left) and early (middle) and late (right) phase fluorescein angiogram (FA) images from the right eye of an 18-year-old man at the initial clinical visit. Temporal retinal vasculitis with vascular sheathing, exudate, retinal hemorrhage, and vitreous haze were noted. The patient had a positive interferon-gamma release assay result and was referred to a pulmonologist, who made a diagnosis of pulmonary TB, confirmed with a positive Mycobacterium tuberculosis culture and high-resolution computed tomography. (B) Fundus photograph and FA image of the right eye showing remission after scatter laser therapy and 6 months of anti-tuberculosis medication.

Fig.┬Ā2
Interferon-gamma release assay (IGRA)-positive rates (A) and false-positive rates (B) among the five age groups examined. Both rates were significantly different among age groups (all p-values <0.001). p-values were obtained by chi-square tests for trend.
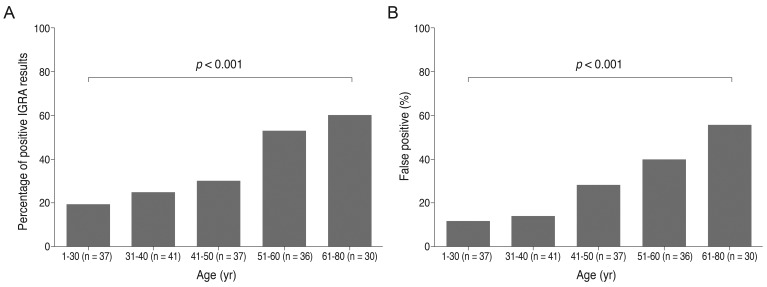
Fig.┬Ā3
Interferon-gamma release assay (IGRA)-positive rates among the four anatomic types of uveitis and between patients with and without retinal vasculitis. (A) The IGRA-positive rates were significantly different among the four anatomic groups (p = 0.001). (B) Patients with retinal vasculitis had higher positive IGRA rate (p < 0.001) than those without retinal vasculitis. p-values were obtained using chi-square tests.
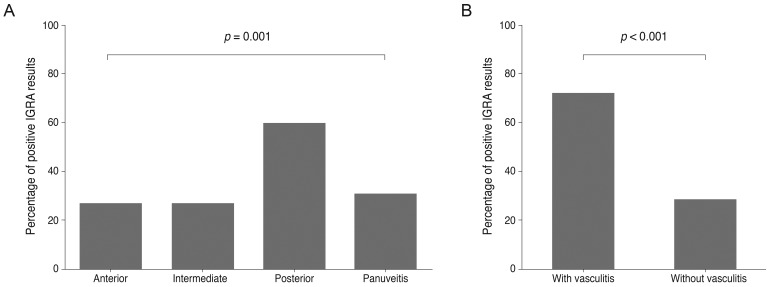
Fig.┬Ā4
Positive predictive values of the interferon-gamma release assay for presumed tuberculosis-related uveitis in patients with intraocular inflammation (A) in the age groups and (B) in the anatomic type subgroups.

- TOOLS



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




