Thyroid-associated ophthalmopathy (TAO) which is characterized by an infiltration of mononuclear cells into orbital tissues, activation of orbital fibroblasts, an increase in production of glycosaminoglycans that cause retention of water and edema, infiltration of extraocular muscles and an expansion of the orbital fat and it is frequently associated with autoimmune toxic goiter due to circulating autoantibodies [
1]. It was shown that an increment in the number of activated T lymphocytes and CD5+ B lymphocytes was detected in patients with TAO, and it may be related to disease severity [
2]. Additionally, promoting inflammatory cytokines in TAO may induce an alteration in the proportion of immune-related cells in the bloodstream [
3], and these changes may provide information about the inflammatory status of TAO patient for the clinician.
Blood cell interactions are crucial in the pathophysiology of immune response and systemic inflammation, and these blood cells including, neutrophils, lymphocytes, monocytes, and platelets, play essential roles in any stage of an autoimmune disease [
4,
5]. Recently, some novel inflammatory markers, including neutrophil-to-lymphocyte ratio (NLR) and platelet-to-lymphocyte ratio (PLR), have been used to assess the severity of inflammation in some autoimmune diseases [
5]. The diagnostic and prognostic values of NLR have been investigated in some ocular diseases such as diabetic retinopathy, age-related macular degeneration, central serous chorioretinopathy, retinal vein occlusion, retinopathy of prematurity, pseudoexfoliation syndrome, keratoconus, optic neuropathy, dry eye disease, and uveitis [
6].
The changes in platelet counts reflect nonspecific inflammatory thrombopoiesis and the release of immune-reactive cells from the bone marrow to the bloodstream in autoimmune diseases [
5]. It was shown that platelet counts might have diagnostic and prognostic value in the indication of autoimmune disease activity and response to anti-inflammatory treatment [
7,
8]. So, NLR and PLR are complementary immuno-inflammatory parameters in the evaluation of any autoimmune disease. Recently, Hu et al. [
9] developed a novel predictable biomarker, systemic immune-inflammatory index (SII), which is a useful inflammation-related indicator to predict clinical outcomes for tumors and other inflammatory diseases, integrates neutrophil, lymphocyte, and platelet counts [
9-
12]. Although only a study has reported that the NLR values of the patients with TAO were significantly higher than those of the controls [
13], there is no study investigating PLR, monocyte-to-lymphocyte ratio (MLR) and SII in patients with TAO.
In this study, we aimed to investigate the blood-count derived immunoinflammatory markers, including NLR, PLR, MLR, and SII in patients with TAO.
Materials and Methods
The study, which had a retrospective and case-control study design, was approved by the Human Research Ethics Committee of Ondokuz May─▒s University (OMU-KAEK-2020/443) and was conducted in accordance with the tenets of the Declaration of Helsinki. Informed consent was obtained from all participants for the use of any clinical data in research. Forty-six patients with TAO and 46 matched healthy controls were included in the study.
Eighty-four patients with TAO still being followed up at the oculoplastic outpatient clinic were retrospectively screened. The patients with TAO who have been diagnosed in our hospital since 2000, and the initial diagnosis have been made or confirmed by the presence of thyroid-stimulating antibody, were included in the study. The complete blood count, thyroid function test, erythrocyte sedimentation rate, and C-reactive protein (CRP) that had been routinely measured during initial diagnosis by the same laboratory, were used in the study. Most of the patients were referred to the ophthalmology department by the endocrinology clinic for ocular examination due to suspicion of TAO or the presence of thyroid-stimulating autoantibodies in serum. The characteristic signs and symptoms of TAO patients were upper eyelid retraction, exophthalmos, lid-lag, scleral show, eyelid swelling and erythema, conjunctival chemosis, caruncle edema, restricted eye movement, and diplopia. TAO was confirmed according to these clinical findings (at least one of them) with or without imaging evidence on computerized tomography or magnetic resonance imaging and the presence of thyroid autoantibodies. TAO activity was evaluated by the clinical activity score according to European Guidelines for European Graves-Basedow Ophthalmopathy. It has seven criteria: spontaneous orbital pain, gaze-evoked orbital pain, eyelid swelling, eyelid erythema, conjunctival redness, chemosis, and caruncle inflammation.
At the time of initial diagnosis, the patients who had used any medicine and had a pregnancy, any disease or disorder, history of radioactive iodine treatment in their medical records, and had incomplete or unreliable medical records were excluded. Age- and sex-matched healthy controls, who did not have any drug use, pregnancy, any disease or disorder, and any familial history of thyroid disease, were recruited through annual health screenings of volunteer health workers.
Blood samples were collected from both the patient and the control group after 12 hours of fasting. All hematologic analyses were performed using an automated haematology analyzer (XN-3000; Sysmex America, Mundelein, IL, USA). Thyroid-stimulating hormone (TSH; normal range, 0.27-4.2 mIU/L), free thyroxine (normal range, 0.93-1.70 ng/dL), free triiodotropin (normal range, 2.0-4.4 ng/dL), anti-thyroid peroxidase antibody (normal range, 0-34 IU/L), anti-thyroglobulin (anti-Tg; normal range, 0-115 IU/L), TSH receptor antibody (normal range, 0-1 IU/L), and CRP (normal range, 0-5 mg/L) were measured by using the electrochemiluminescence method using Roche Cobas 8000 analyzer (Roche Diagnostic). The NLR, PLR, and MLR were calculated as the ratio of the neutrophil-to-lymphocyte, platelet-to-lymphocyte, and monocyte-to-lymphocyte. SII was calculated as (absolute count of platelets ├Ś absolute count of neutrophils)/absolute count of lymphocytes [
9-
12].
The current data of the patients including, visual acuity, intraocular pressure, exophthalmometry, lid retraction, restrictive myopathy, corneal involvement and history of extraocular muscle involvement and optic nerve involvement, and laboratory parameters at the time of initial diagnosis were recorded for statistical analysis.
Intravenous steroid treatment was administered at least once to the clinically active patients (clinical activity score >3) having commonly soft tissue inflammation accompanied with myositis (n = 12) or optic nerve compression (n = 2) during the follow-up period. In patients with optic nerve compression, intravenous steroids were administered at a high dose (1 g methylprednisolone) for three days, followed by oral prednisolone (totally 4.5 g). None of the patients needed any decompression surgery. The patients with myositis received intravenous 500 mg methylprednisolone once a week for six weeks, followed by oral prednisolone treatment (totally 4.5 g). None of the patients in the study received radiotherapy or any other immunosuppressant therapy except systemic steroid treatment during the follow-up period.
Data were statistically analyzed using IBM SPSS Statistics ver. 22 (IBM Corp., Armonk, NY, USA). Mann-Whitney U-test was performed to compare the values of the patients with TAO and the controls. Receiver operating characteristics curve analysis was used for laboratory parameters in predicting TAO and subsequent steroid requirement. SpearmanŌĆÖs correlation test was conducted to analyze the association of the immunoinflammatory parameters with the clinical and ocular variables among TAO eyes. Logistic regression analyses were performed to identify inflammatory risk factors for TAO patients. A p-value <0.05 indicated statistical significance.
Results
The mean age of TAO (27 female and 19 male) and control groups (27 female and 19 male) were 45.83 ┬▒ 15.14 years (range, 23-70 years) and 46.09 ┬▒ 15.42 years (range, 23-70 years), respectively. The characteristics of TAO and control groups are summarized in
Table 1, and the laboratory results of these groups were compared in
Table 2.
There were statistically significant differences in NLR, PLR, SII, and lymphocyte count between the TAO and the control groups, while no significant difference in MLR was found between them. Mean NLR, PLR and SII were significantly lower in the patients with TAO than in the controls, while the mean value of lymphocyte count in the TAO group was significantly higher than in the control group (
p < 0.05). The area under the receiver operating characteristic curve values of NLR, SII, count of lymphocytes, and PLR to distinguish TAO from healthy subjects were 0.68, 0.65, 0.67, and 0.64, respectively (
Table 3 and
Fig. 1A-1D).
In logistic regression analysis, any blood count-derived parameters were not associated with the risks of having proptosis, lid retraction, myositis, restrictive myopathy, systemic steroid need, corneal involvement, and optic neuropathy during the follow-up period (
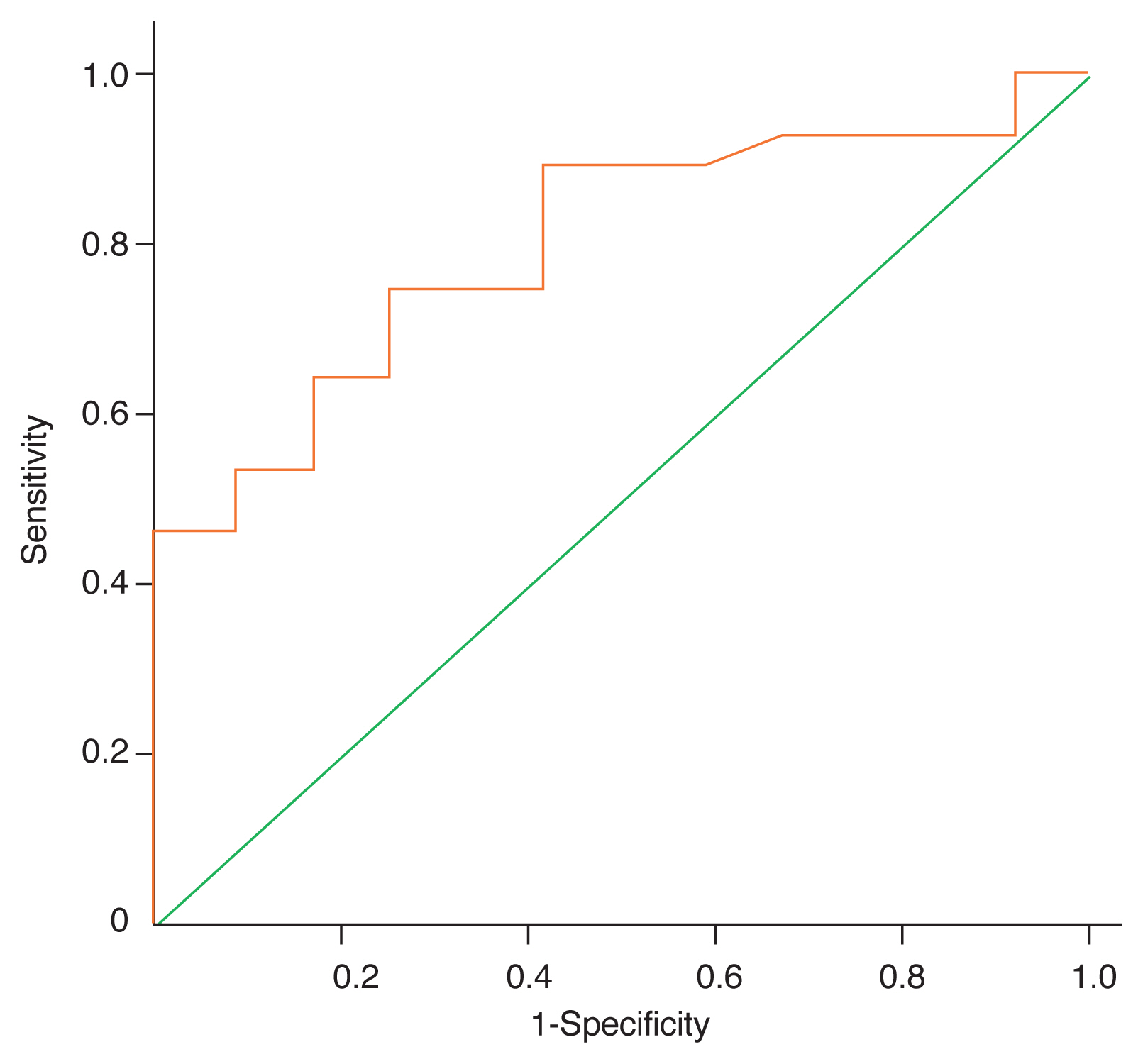
p > 0.05 for all). The patients, who needed systemic treatment due to TAO, had significantly lower platelet count and PLR at the time of initial diagnosis when compared to the TAO patients who did not need any systemic treatment during the follow-up period (
Table 4). Platelet count and PLR were significantly correlated to erythrocyte sedimentation rate (r = 0.75,
p = 0.008 and r = 0.70,
p = 0.016, respectively), while there was no significant association between the blood count-derived parameters and CRP (
p > 0.05). In the logistic regression model, platelet count was significantly associated with the requirement of steroid use due to TAO (╬▓ = ŌłÆ0.02,
p = 0.03). The best cut-off value of initial platelet count to determine the subsequent need for steroid use during the follow-up in TAO patients was 194,000, with 89.3% sensitivity and 59.1% specificity (area under the receiver operating characteristic curve, 0.80;
p = 0.003; 95% confidence interval, 0.66-0.94) (
Fig. 2).
Additionally, NLR and SII had significant negative correlation with anti-Tg (r = ŌłÆ0.34, p = 0.04 and r = ŌłÆ0.37, p = 0.03), while the platelet, neutrophil and lymphocyte counts were not specifically correlated to anti-Tg (p > 0.05). There was not any association between any blood parameters and TSH, free triiodotropin, and free thyroxine.
Discussion
TAO is an infiltrative and lymphocyte-dependent autoimmune disease, in which activated T lymphocytes and transitional and pre-naïve mature B lymphocytes are responsible for the autoimmune process [
13]. During this process, amounts of circulating mononuclear cell subpopulations may be altered by secreted cytokines and mediators, and cell to cell interactions and may give information about the disease activity or prognosis [
4,
14,
15].
Blood count-derived immunoinflammatory biomarkers including NLR, PLR, MLR, and SII are inexpensive and easily accessible predictors to evaluate systemic inflammation, and they have been identified to be associated with the prognosis and the activity of many cardiovascular, oncologic and autoimmune diseases [
5,
6,
11,
16]. NLR is the most investigated inflammatory parameter in ocular diseases, and higher NLR level was found to be associated with the presence of diabetic retinopathy, age-related macular degeneration, acute optic neuritis, retinal vein occlusion, progressive keratoconus, uveitis, dry eye, pseudoexfoliation and primary open-angle glaucoma [
6]. In literature, only one study evaluating the relationship between NLR and TAO, in which the values of NLR were found to be higher in patients with TAO than in controls [
17]. In contrast to this study, we have found that NLR was significantly lower in the group of TAO when comparing with age- and sex-matched healthy controls. In both studies, the number of lymphocytes was higher in the TAO group than in the controls, but the number of neutrophils was different between the studies. This difference may be due to the reason that the previous study has more active TAO patients than the present study has. It was reported that lymphocyte count was slightly increased, and granulocytes (neutrophils mainly) was considerably decreased in GravesŌĆÖ disease [
2,
15,
18,
19]. Although the lymphocyte and the neutrophil counts were not correlated to any TAO related antibody, NLR was significantly correlated to anti-Tg. However, the logistic regression analysis showed that NLR has no prognostic significance for clinical outcomes in TAO patients. Additionally, we found that the best cut-off value of NLR to distinguish TAO patients and healthy subjects was 2.11, with 76.2% sensitivity and 56.2% specificity.
Platelets have a critical role in regulating inflammatory reactions and immune responses by the modulation of lymphocyte functioning. In the present study, the TAO group had significantly lower PLR than healthy controls, and the best cut-off value of PLR to distinguish TAO patients and healthy subjects was 126.09, with 64.2% sensitivity and 50.1% specificity. Additionally, the initial platelet count was significantly related to the need for systemic steroid treatment due to TAO in the logistic regression model (╬▓ = ŌłÆ0.02,
p = 0.03). Even though the difference in PLR is mainly due to the difference in lymphocyte counts between the groups in the study, the role of platelets in autoimmune response is undeniable. The interactions of platelets with T-lymphocytes decreases lymphocyte proliferation and reduces the amount of pro-inflammatory cytokines, such as interferon-alpha, tumor necrosis factor-alpha, and interleukin (IL) 17, and increase in anti-inflammatory cytokines, such as IL-10 [
5,
20]. These cytokines are critical for regulation and differentiation of T helper-17 cells which have been linked to various autoimmune diseases, such as rheumatoid arthritis, psoriasis, inflammatory bowel disease, multiple sclerosis, and TAO [
4]. Kim et al. [
21] found that the serum levels of IL-17 in active TAO patients were higher than those in inactive TAO patients, and serum IL-17 concentration significantly correlated with the disease activity. In the pathogenesis of TAO, platelets may produce aggregations with lymphocytes to promote lymphocyte rolling and adhesion by P-selectin bridging [
22], enhancing lymphocyte adhesion to the endothelium [
23] and facilitating lymphocyte homing [
24]. It may be thought that increased interactions between lymphocytes and platelets may cause significant augmentation in lymphocyte infiltration through the tissue by these lymphocyte-platelet aggregates, and the infiltration of immune reactive lymphocytes may aggravate the disease activity during the following periods of the disease.
Although thyroid hormone levels were previously thought to have a lowering effect on the platelet count in autoimmune thyroid disease [
25-
27], many studies show that autoantibody against platelets may occur in patients with the disease [
28-
31]. In the present study, the platelet count and PLR were significantly lower in the group who needed systemic steroid treatment due to TAO during the follow-up period than in the no treatment-needed group, although there was no significant difference in thyroid function test between these groups (
Table 4). Recently, Ito et al. [
29] reported that platelet-associated immunoglobulin G levels in patients were higher in GravesŌĆÖ disease, and it was hypothesized that GravesŌĆÖ disease triggers some autoimmune processes leading to the production of antibodies against the platelets [
28-
31]. Thus, the platelet count at the initial diagnosis may help predict the severity and extent of the autoimmunity in autoimmune thyroid disease, and it may be speculated that baseline lower platelet count may be linked to TAO severity during the follow-up and may have prognostic significance in the patients of TAO even if it is within normal limits. The best cut-off value of initial platelet count to determine the subsequent need for systemic steroid therapy during the follow-up in TAO patients was 194,000, with 89.3% sensitivity and 59.1% specificity (
Fig. 2). The patients with relatively low platelet levels at the time of initial diagnosis can be closely monitored, as they are more likely to develop ophthalmopathy that will require steroid use in the future.
Although the human CD34+ fibrocytes are circulating monocyte lineage progenitor cells and recently been implicated in TAO [
32], we have not found any difference in monocyte count and MLR between the groups of TAO and control.
In autoimmune diseases, PLR values should be evaluated along with the values of NLR and other inflammatory markers, which may allow of overviewing the whole picture of the immunoinflammatory status of the patients [
5]. SII is recently developed to evaluate the immunoinflammatory activity by combined analysis of neutrophil, lymphocyte, and platelet counts together and to predict clinical outcomes for tumors, cardiovascular and other inflammatory diseases [
9-
12,
16]. Recently, Tang et al. [
33] reported that elevated NLR and SII might serve as readily available inflammatory predictors in POAG patients. To the best of our knowledge, this is the first study investigating MLR, PLR, and SII in patients with TAO, and we have found that SII was significantly lower in the TAO group than in control subjects and the best cut-off value of SII to discriminate TAO patients and healthy subjects was 550.77, with 67.7% sensitivity and 52.6% specificity.
This study may have some limitations. The retrospective design of the study may have caused selection bias, and it may have a relatively small sample to show precise predictive values of blood count-derived parameters. Although these results may not be representative of all patients with TAO, the results of the study may provide critical information to clinicians when evaluating the immune-inflammatory status of a newly diagnosed patient with autoimmune thyroid disease.
In conclusion, the blood count-derived markers are readily available laboratory parameters that may be used in the clinical evaluation of patients with TAO. NLR, PLR, and SII may serve as potential inflammatory biomarkers in the detection of the TAO, and relatively lower platelet count at initial diagnosis may have a prognostic effect in the patients with TAO. Further prospective longitudinal studies with a larger sample size should be needed to investigate the prognostic and predictive values of the novel immune-inflammatory biomarkers in the patients of TAO.
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
Fig.┬Ā1
Receiver operating characteristics curve analysis of (A) neutrophil-to-lymphocyte ratio, (B) platelet-to-lymphocyte ratio, (C) systemic immune-inflammatory index, and (D) lymphocyte count for discrimination between healthy subjects and thyroid-associated ophthalmopathy.

Fig.┬Ā2
Receiver operating characteristics curve analysis of platelets for detection the subsequent systemic steroid need due to thyroid-associated ophthalmopathy during the follow-up period.
Table┬Ā1
The characteristics of(TA) and control groups are summarize
|
TAO group (n = 46) |
Control group (n = 46) |
p-value |
|
Age (yr) |
45.83 ┬▒ 15.14 (23-70) |
46.09 ┬▒ 15.42 (23-70) |
0.9 |
|
Sex (female: male) |
27: 19 |
27: 19 |
- |
|
Body mass index (kg/m2) |
24.01 ┬▒ 3.41 (20.5-31.25) |
24.34 ┬▒ 3.14 (21.09-32.03) |
0.7 |
|
Duration of follow-up (yr) |
9.5 ┬▒ 6.6 (1-15) |
- |
|
|
Clinical activity score |
1.46 ┬▒ 1.34 (0-4) |
- |
- |
|
Exophthalmometry (mm) |
20.27 ┬▒ 3.66 (15-27.5) |
- |
- |
|
Proptosis (unilateral: bilateral: no) |
9: 20: 17 |
- |
- |
|
Lid retraction (unilateral: bilateral: no) |
14: 20: 12 |
- |
- |
|
Positive history of myositis |
16 |
- |
- |
|
Patients needing systemic treatment |
14 |
- |
- |
|
Restrictive myopathy |
10 |
- |
- |
|
Corneal involvement |
4 |
- |
- |
|
Optic nerve involvement |
2 |
- |
- |
Table┬Ā2
The laboratory findings of TAO and the control group
|
Parameter |
TAO group (n = 46) |
Control group (n = 46) |
p-value |
|
TSH (mIU/L) |
1.27 ┬▒ 2.25 (0.00-10.26) |
3.01 ┬▒ 1.01 (1.1-4.22) |
<0.001*
|
|
fT3 (ng/dL) |
7.41 ┬▒ 7.09 (0.1-30.56) |
3.3 ┬▒ 0.57 (2.4-4.54) |
<0.001*
|
|
fT4 (ng/dL) |
2.12 ┬▒ 2.18 (0.37-13.00) |
1.1 ┬▒ 0.25 (0.85-2.12) |
<0.001*
|
|
Anti-TPO (IU/mL) |
183.74 ┬▒ 171.5 (5.0-509.6) |
- |
- |
|
Anti-Tg (IU/mL) |
177.18 ┬▒ 174.96 (10.0-716.7) |
- |
- |
|
WBC (109/L) |
7.71 ┬▒ 1.96 (3.14-13.22) |
7.38 ┬▒ 1.35 (4.51-9.88) |
0.2 |
|
Neutrophils (109/L) |
4.57 ┬▒ 1.71 (2.21-10.20) |
4.64 ┬▒ 1.96 (2.62-12.96) |
0.59 |
|
Lymphocytes (109/L) |
2.56 ┬▒ 0.73 (1.48-4.20) |
2.08 ┬▒ 0.62 (0.83-3.41) |
0.01*
|
|
Monocytes (109/L) |
0.48 ┬▒ 0.18 (0.10-1.00) |
0.48 ┬▒ 0.18 (0.21-1.12) |
0.45 |
|
Platelets (109/L) |
264.57 ┬▒ 68.72 (154-435) |
260.97 ┬▒ 63.74 (169-493) |
0.55 |
|
NLR |
1.82 ┬▒ 0.65 (0.88-4.30) |
2.41 ┬▒ 1.15 (1.06-5.93) |
0.01*
|
|
PLR |
104.58 ┬▒ 37.1 (45.05-179.0) |
134.02 ┬▒ 44.25 (74.13-262.65) |
0.03*
|
|
MLR |
0.20 ┬▒ 0.09 (0.03-0.48) |
0.24 ┬▒ 0.09 (0.13-0.40) |
0.15 |
|
SII |
470.30 ┬▒ 263.45 (191.89-859.05) |
607.94 ┬▒ 258.48 (223.86-1092.3) |
0.02*
|
|
ESR (mm/hr) |
24.13 ┬▒ 18.25 (8-71) |
- |
- |
|
CRP (mg/L) |
0.99 ┬▒ 0.57 (0.20-2.21) |
- |
- |
Table┬Ā3
Receiver operating characteristics curve analysis for blood count-derived immunoinflammatory biomarkers and lymphocyte count in predicting thyroid-associated ophthalmopathy
|
Parameter |
Best cut-off value |
AUROC |
p-value |
Sensitivity (%) |
Specificity (%) |
95% CI |
|
NLR |
2.11 |
0.68 |
0.011*
|
76.2 |
52.6 |
0.55-0.80 |
|
PLR |
126.09 |
0.64 |
0.03*
|
64.2 |
50.1 |
0.51-0.76 |
|
SII |
550.77 |
0.65 |
0.03*
|
67.7 |
52.6 |
0.52-0.78 |
|
MLR |
0.198 |
0.58 |
0.21 |
55.2 |
52.6 |
0.45-0.72 |
|
L (109/L) |
2.15 |
0.67 |
0.012*
|
66.7 |
55.8 |
0.55-0.80 |
Table┬Ā4
Comparison of the initial blood parameters between the patients who needed systemic steroid treatment due to TAO and the TAO patients who did not need any systemic treatment during the follow-up
|
Parameter |
Systemic treatment-needed group (n = 14) |
No treatment-needed group (n = 32) |
p-value |
|
WBC (109/L) |
8.18 ┬▒ 1.94 (5.30-12.94) |
7.58 ┬▒ 2.09 (3.14-13.22) |
0.33 |
|
Neutrophils (109/L) |
4.75 ┬▒ 1.56 (3.20-9.59) |
4.37 ┬▒ 1.60 (2.21-10.20) |
0.34 |
|
Lymphocytes (109/L) |
2.51 ┬▒ 0.66 (1.50-3.75) |
2.60 ┬▒ 0.71 (1.48-4.20) |
0.48 |
|
Monocytes (109/L) |
0.50 ┬▒ 0.15 (0.31-0.91) |
0.46 ┬▒ 0.19 (0.10-1.00) |
0.06 |
|
Platelets (109/L) |
214.38 ┬▒ 46.18 (154-300) |
285.61 ┬▒ 66.08 (158-435) |
0.001*
|
|
NLR |
1.96 ┬▒ 0.64 (1.16-3.75) |
1.77 ┬▒ 0.76 (0.88-4.30) |
0.15 |
|
PLR |
99.65 ┬▒ 35.69 (45.58-176.85) |
110.93 ┬▒ 33.49 (45.05-179.0) |
0.02*
|
|
MLR |
0.24 ┬▒ 0.09 (0.1-0.48) |
0.18 ┬▒ 0.08 (0.03-0.48) |
0.06 |
|
SII |
487.14 ┬▒ 300.14 (191.89-845.35) |
463.06 ┬▒ 186.69 (217.14-859.05) |
0.49 |
|
ESR (mm/hr) |
26.40 ┬▒ 25.33 (9-70) |
23.75 ┬▒ 21.13 (8-71) |
0.84 |
|
CRP (mg/L) |
0.73 ┬▒ 0.46 (0.20-1.00) |
1.13 ┬▒ 0.56 (0.55-2.21) |
0.43 |
|
TSH (mIU/L) |
1.60 ┬▒ 2.24 (0.0-7.03) |
1.23 ┬▒ 2.28 (0.0-10.26) |
0.76 |
|
fT3 (ng/dL) |
8.02 ┬▒ 7.14 (0.66-25.0) |
6.88 ┬▒ 6.87 (0.1-30.56) |
0.91 |
|
fT4 (ng/dL) |
2.10 ┬▒ 1.45 (0.77-5.09) |
2.13 ┬▒ 2.35 (0.37-13.00) |
0.90 |
|
Anti-TPO (IU/mL) |
252.58 ┬▒ 206.35 (8.57-502.5) |
158.81 ┬▒ 142.74 (5.0-509.6) |
0.36 |
|
Anti-Tg (IU/mL) |
190.40 ┬▒ 119.19 (10.0-716.7) |
160.72 ┬▒ 190.71 (10.0-708.4) |
0.67 |
References
1. Yamada M, Li AW, Wall JR. Thyroid-associated ophthalmopathy: clinical features, pathogenesis, and management.
Crit Rev Clin Lab Sci 2000;37:523-49.


2. Van der Weerd K, Van Hagen PM, Schrijver B, et al. The peripheral blood compartment in patients with GravesŌĆÖ disease: activated T lymphocytes and increased transitional and pre-naive mature B lymphocytes.
Clin Exp Immunol 2013;174:256-64.



3. Tabarkiewicz J, Pogoda K, Karczmarczyk A, et al. The role of IL-17 and Th17 lymphocytes in autoimmune diseases.
Arch Immunol Ther Exp (Warsz) 2015;63:435-49.



4. Kristensen B. Regulatory B and T cell responses in patients with autoimmune thyroid disease and healthy controls.
Dan Med J 2016;63:B5177.

5. Gasparyan AY, Ayvazyan L, Mukanova U, et al. The platelet-to-lymphocyte ratio as an inflammatory marker in rheumatic diseases.
Ann Lab Med 2019;39:345-57.



6. Kurtul BE, Ozer PA. Neutrophil-to-lymphocyte ratio in ocular diseases: a systematic review.
Int J Ophthalmol 2019;12:1951-8.



7. Gasparyan AY, Sandoo A, Stavropoulos-Kalinoglou A, Kitas GD. Mean platelet volume in patients with rheumatoid arthritis: the effect of anti-TNF-╬▒ therapy.
Rheumatol Int 2010;30:1125-9.


8. Abdel Galil SM, Edrees AM, Ajeeb AK, et al. Prognostic significance of platelet count in SLE patients.
Platelets 2017;28:203-7.


9. Hu B, Yang XR, Xu Y, et al. Systemic immune-inflammation index predicts prognosis of patients after curative resection for hepatocellular carcinoma.
Clin Cancer Res 2014;20:6212-22.


10. Ma M, Yu N, Wu B. High systemic immune-inflammation index represents an unfavorable prognosis of malignant pleural mesothelioma.
Cancer Manag Res 2019;11:3973-9.


11. Jomrich G, Gruber ES, Winkler D, et al. Systemic Immune-Inflammation Index (SII) predicts poor survival in pancreatic cancer patients undergoing resection.
J Gastrointest Surg 2020;24:610-8.


12. Furuncuoglu Y, Tulgar S, Dogan AN, et al. How obesity affects the neutrophil/lymphocyte and platelet/lymphocyte ratio, systemic immune-inflammatory index and platelet indices: a retrospective study.
Eur Rev Med Pharmacol Sci 2016;20:1300-6.

13. Klecha AJ, Barreiro Arcos ML, Frick L, et al. Immune-endocrine interactions in autoimmune thyroid diseases.
Neuroimmunomodulation 2008;15:68-75.


14. Tanaka T. Changes of peripheral mononuclear cell subpopulations in autoimmune thyroid diseases.
Nihon Naibunpi Gakkai Zasshi 1985;61:711-23.


15. Tomczynska M, Saluk-Bijak J. The mutual cooperation of blood platelets and lymphocytes in the development of autoimmune thyroid diseases.
Acta Biochim Pol 2018;65:17-24.


16. Huang J, Zhang Q, Wang R, et al. Systemic Immune-Inflammatory Index predicts clinical outcomes for elderly patients with acute myocardial infarction receiving percutaneous coronary intervention.
Med Sci Monit 2019;25:9690-701.



17. Celik T. Neutrophil-to-lymphocyte ratio in thyroid ophthalmopathy.
Bratisl Lek Listy 2017;118:495-8.


18. Eakin DL, Peake RL, Weiss GB. Effect of therapy on the neutropenia of hyperthyroidism.
South Med J 1983;76:335-7.


19. Taskaldiran I, Omma T, Onder CE, et al. Neutrophil-to-lymphocyte ratio, monocyte-to-lymphocyte ratio, and platelet-tolymphocyte ratio in different etiological causes of thyrotoxicosis.
Turk J Med Sci 2019;49:1687-92.


20. Zamora C, Canto E, Nieto JC, et al. Binding of platelets to lymphocytes: a potential anti-inflammatory therapy in rheumatoid arthritis.
J Immunol 2017;198:3099-108.


21. Kim SE, Yoon JS, Kim KH, Lee SY. Increased serum interleukin-17 in GravesŌĆÖ ophthalmopathy.
Graefes Arch Clin Exp Ophthalmol 2012;250:1521-6.


22. Lalor P, Nash GB. Adhesion of flowing leucocytes to immobilized platelets.
Br J Haematol 1995;89:725-32.


23. Diacovo TG, Catalina MD, Siegelman MH, von Andrian UH. Circulating activated platelets reconstitute lymphocyte homing and immunity in L-selectin-deficient mice.
J Exp Med 1998;187:197-204.



24. Kuznik BI, Vitkovsky YA, Gvozdeva OV, et al. Lymphocyte-platelet crosstalk in GravesŌĆÖ disease.
Am J Med Sci 2014;347:206-10.


25. Kurata Y, Nishioeda Y, Tsubakio T, Kitani T. Thrombocytopenia in GravesŌĆÖ disease: effect of T3 on platelet kinetics.
Acta Haematol 1980;63:185-90.


26. Masunaga R, Nagasaka A, Nakai A, et al. Alteration of platelet aggregation in patients with thyroid disorders.
Metabolism 1997;46:1128-31.


27. Okamoto A, Taniguchi K, Kajita Y. Improvement of thrombocytopenia with normalization of thyroid function in a patient with Graves disease.
Rinsho Ketsueki 2006;47:536-8.

28. Hymes K, Blum M, Lackner H, Karpatkin S. Easy bruising, thrombocytopenia, and elevated platelet immunoglobulin G in GravesŌĆÖ disease and HashimotoŌĆÖs thyroiditis.
Ann Intern Med 1981;94:27-30.


29. Ito S, Fujiwara SI, Murahashi R, et al. Clinical association between thyroid disease and immune thrombocytopenia.
Ann Hematol 2021;100:345-52.


30. Verges B, Giroud-Baleydier F, Olsson O, et al. Association of BasedowŌĆÖs disease and autoimmune thrombopenic purpura. No effect of hyperthyroidism on antiplatelet autoimmunity.
Rev Med Interne 1989;10:565-9.

31. Lhotta K, Zitt E, Sprenger-Mahr H, et al. Treatment of concurrent thrombotic thrombocytopenic purpura and GravesŌĆÖ disease: a report on two cases.
Case Rep Endocrinol 2018;2018:5747969.



32. Fernando R, Grisolia ABD, Lu Y, et al. Slit2 modulates the inflammatory phenotype of orbit-infiltrating fibrocytes in GravesŌĆÖ disease.
J Immunol 2018;200:3942-9.



33. Tang B, Li S, Han J, et al. Associations between blood cell profiles and primary open-angle glaucoma: a retrospective case-control study.
Ophthalmic Res 2020;63:413-22.









 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




